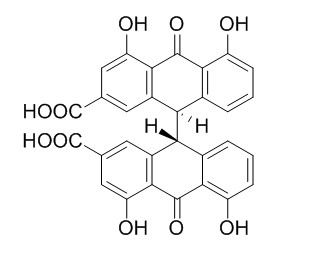
Sennidin A
Sennidin A, has two hydroxyanthraquinone-like moieties, exerts inhibition on NS3 helicase with IC50 values of 0.8 uM. Sennidin A in combination with necrosis inducing drugs/therapies may generate synergetic tumoricidal effects on solid malignancies by means of primary debulking and secondary cleansing process. It stimulates glucose incorporation in the phosphatidylinositol 3-kinase (PI3K)- and Akt-dependent in rat adipocytes, but in the IR/IRS1-independent manner.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Phytother Res.2022, ptr.7573.
iScience.2024, 4790628.
Oncol Rep.2019, 41(4):2453-2463
Appl. Sci.2022, 12(17), 8646.
Fitoterapia.2024, 177:106138.
Biomolecules.2024, 14(5):589.
Molecules.2020, 25(15):3353.
Food Chem.2021, 337:128023.
Dent Mater J. 2024, dmj.2023-294.
Food Bioscience2023, 56:103311.
Related and Featured Products
Oncotarget. 2014 May 30;5(10):2934-46.
Necrosis targeted combinational theragnostic approach using radioiodinated Sennidin A in rodent tumor models.[Pubmed:
24931286]
Residual cancer cells and subsequent tumor relapse is an obstacle for curative cancer treatment. Tumor necrosis therapy (TNT) has recently been developed to cause residual tumor regression or destruction.
METHODS AND RESULTS:
Here, we exploited the avidity of the Sennidin A (SA) tracer and radioiodinated SA (¹³¹I-SA) to necrotic tumors in order to further empower TNT. We showed high uptake and prolonged retention of SA in necrotic tumors and a quick clearance in other non-targeted tissues including the liver. On SPECT-CT images, tumor mass appeared persistently as a hotspot. Based on the prominent targetability of ¹³¹I-SA to the tumor necrosis, we designed a combinational theragnostic modality. The vascular disrupting agent (VDA) combretastatin A4 phosphate (CA4P) was used to cause massive tumor necrosis, which formed the target of ¹³¹I-SA that subsequently killed the residual tumor cells by cross-fire irradiation of beta particles.
Consequently, ¹³¹I-SA combined with CA4P significantly inhibited tumor growth, extended tumor doubling time and prolonged mean animal survival.
CONCLUSIONS:
In conclusion, ¹³¹I-SA in combination with necrosis inducing drugs/therapies may generate synergetic tumoricidal effects on solid malignancies by means of primary debulking and secondary cleansing process.
Int J Mol Sci. 2015 Aug; 16(8): 18439–53.
Identification of Hydroxyanthraquinones as Novel Inhibitors of Hepatitis C Virus NS3 Helicase[Pubmed:
26262613]
Hepatitis C virus (HCV) is an important etiological agent of severe liver diseases, including cirrhosis and hepatocellular carcinoma. The HCV genome encodes nonstructural protein 3 (NS3) helicase, which is a potential anti-HCV drug target because its enzymatic activity is essential for viral replication. Some anthracyclines are known to be NS3 helicase inhibitors and have a hydroxyanthraquinone moiety in their structures; mitoxantrone, a hydroxyanthraquinone analogue, is also known to inhibit NS3 helicase.
METHODS AND RESULTS:
Therefore, we hypothesized that the hydroxyanthraquinone moiety alone could also inhibit NS3 helicase. Here, we performed a structure–activity relationship study on a series of hydroxyanthraquinones by using a fluorescence-based helicase assay. Hydroxyanthraquinones inhibited NS3 helicase with IC50 values in the micromolar range. The inhibitory activity varied depending on the number and position of the phenolic hydroxyl groups, and among different hydroxyanthraquinones examined, 1,4,5,8-tetrahydroxyanthraquinone strongly inhibited NS3 helicase with an IC50 value of 6 μM. Furthermore, hypericin and Sennidin A, which both have two hydroxyanthraquinone-like moieties, were found to exert even stronger inhibition with IC50 values of 3 and 0.8 μM, respectively.
CONCLUSIONS:
These results indicate that the hydroxyanthraquinone moiety can inhibit NS3 helicase and suggest that several key chemical structures are important for the inhibition.
Life Sci. 2006 Aug 8;79(11):1027-33.
Sennidin stimulates glucose incorporation in rat adipocytes.[Pubmed:
16603199]
A novel small molecule compound which exerts insulin mimetic is desirable. Dozens of natural products that have quinone, naphthoquinone, or anthraquinone structure, were tested by a glucose incorporation assay.
METHODS AND RESULTS:
We found that Sennidin A, anthraquinone derivative, stimulated glucose incorporation to near level of maximal insulin-stimulated and sennidin B, a stereoisomer of Sennidin A, also stimulated, but the activity of sennidin B was lower than Sennidin A. Sennidin A-stimulated glucose incorporation was completely inhibited by wortmannin. Sennidin A did not induce tyrosine phosphorylation of insulin receptor (IR) and insulin receptor substrate-1 (IRS-1), but induced phosphorylation of Akt and glucose transporter 4 (GLUT4) translocation.
CONCLUSIONS:
Our results suggest that in rat adipocytes, Sennidin A stimulates glucose incorporation in the phosphatidylinositol 3-kinase (PI3K)- and Akt-dependent, but in the IR/IRS1-independent manner.
Appl Environ Microbiol. 1994 Mar;60(3):1041-3.
Isolation of a human intestinal anaerobe, Bifidobacterium sp. strain SEN, capable of hydrolyzing sennosides to sennidins.[Pubmed:
8161172]
METHODS AND RESULTS:
A strictly anaerobic bacterium capable of metabolizing sennosides was isolated from human feces and identified as Bifidobacterium sp., named strain SEN. The bacterium hydrolyzed sennoside A and sennidin B to Sennidin A and sennidin B via Sennidin A and B 8-monoglucosides, respectively.
CONCLUSIONS:
Among nine species of Bifidobacterium having beta-glucosidase activity, only Bifidobacterium dentium and B. adolescentis metabolized sennoside B to sennidin B, suggesting that the sennoside-metabolizing bacteria produce a novel type of beta-glucosidase capable of hydrolyzing sennosides to sennidins.