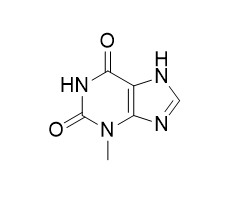
3-Methylxanthine
3-Methylxanthine inhibited xanthine crystallization, it could protect patients with xanthinuria from the development of renal xanthine calculi.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
J Health Sci Med Res.2023, 31584.
Clin Transl Med.2021, 11(5):e392.
Anticancer Res.2024, 44(3):1033-1044.
ACS Synth Biol.2022, 11(10):3296-3304.
Expert Opin Ther Targets.2024, :1-11.
Evid Based Complement Alternat Med.2022, 2022:3483511
Oxid Med Cell Longev2020, 12
Universiti Tunku Aboul Rahman2023, 6263.
J Drug Target.2016, 24:1-28
Plos One.2020, 10.1371
Related and Featured Products
PLoS One. 2018 Aug 29;13(8):e0198881.
Xanthine urolithiasis: Inhibitors of xanthine crystallization.[Pubmed:
30157195 ]
To identify in vitro inhibitors of xanthine crystallization that have potential for inhibiting the formation of xanthine crystals in urine and preventing the development of the renal calculi in patients with xanthinuria.
METHODS AND RESULTS:
The formation of xanthine crystals in synthetic urine and the effects of 10 potential crystallization inhibitors were assessed using a kinetic turbidimetric system with a photometer. The maximum concentration tested for each compound was: 20 mg/L for 3-Methylxanthine (3-MX); 40 mg/L for 7-methylxanthine (7-MX), 1-methylxanthine (1-MX), theobromine (TB), theophylline, paraxanthine, and caffeine; 45 mg/L for 1-methyluric acid; 80 mg/L for 1,3-dimethyluric acid; and 200 mg/L for hypoxanthine. Scanning electron microscopy was used to examine the morphology of the crystals formed when inhibitory effects were observed.
Only 7-MX, 3-MX, and 1-MX significantly inhibited xanthine crystallization at the tested concentrations. Mixtures of inhibitors had an additive effect rather than a synergistic effect on crystallization.
CONCLUSIONS:
Two of the inhibitors identified here-7-MX and 3-MX-are major metabolites of TB. In particular, after TB consumption, 20% is excreted in the urine as TB, 21.5% as 3-MX, and 36% as 7-MX. Thus, consumption of theobromine could protect patients with xanthinuria from the development of renal xanthine calculi. Clinical trials are necessary to demonstrate these effects in vivo.
Microb Cell Fact. 2015 Dec 21;14:203.
Direct conversion of theophylline to 3-methylxanthine by metabolically engineered E. coli.[Pubmed:
26691652 ]
Methylxanthines are natural and synthetic compounds found in many foods, drinks, pharmaceuticals, and cosmetics. Aside from caffeine, production of many methylxanthines is currently performed by chemical synthesis. This process utilizes many chemicals, multiple reactions, and different reaction conditions, making it complicated, environmentally dissatisfactory, and expensive, especially for monomethylxanthines and paraxanthine. A microbial platform could provide an economical, environmentally friendly approach to produce these chemicals in large quantities. The recently discovered genes in our laboratory from Pseudomonas putida, ndmA, ndmB, and ndmD, provide an excellent starting point for precisely engineering Escherichia coli with various gene combinations to produce specific high-value paraxanthine and 1-, 3-, and 7-methylxanthines from any of the economical feedstocks including caffeine, theobromine or theophylline. Here, we show the first example of direct conversion of theophylline to 3-Methylxanthine by a metabolically engineered strain of E. coli.
METHODS AND RESULTS:
Here we report the construction of E. coli strains with ndmA and ndmD, capable of producing 3-Methylxanthine from exogenously fed theophylline. The strains were engineered with various dosages of the ndmA and ndmD genes, screened, and the best strain was selected for large-scale conversion of theophylline to 3-Methylxanthine. Strain pDdA grown in super broth was the most efficient strain; 15 mg/mL cells produced 135 mg/L (0.81 mM) 3-Methylxanthine from 1 mM theophylline. An additional 21.6 mg/L (0.13 mM) 1-methylxanthine were also produced, attributed to slight activity of NdmA at the N 3 -position of theophylline. The 1- and 3-Methylxanthine products were separated by preparative chromatography with less than 5% loss during purification and were identical to commercially available standards. Purity of the isolated 3-Methylxanthine was comparable to a commercially available standard, with no contaminant peaks as observed by liquid chromatography-mass spectrophotometry or nuclear magnetic resonance.
CONCLUSIONS:
We were able to biologically produce and separate 100 mg of highly pure 3-Methylxanthine from theophylline (1,3-dimethylxanthine). The N-demethylation reaction was catalyzed by E. coli engineered with N-demethylase genes, ndmA and ndmD. This microbial conversion represents a first step to develop a new biological platform for the production of methylxanthines from economical feedstocks such as caffeine, theobromine, and theophylline.