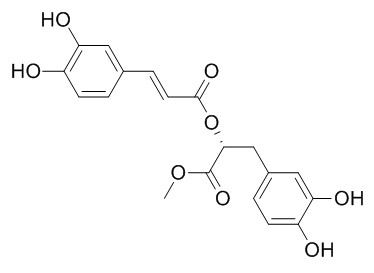
Methyl rosmarinate
Methyl rosmarinate shows antioxidative, and antifungal activities. It has inhibitory activities against tyrosinase, α-glucosidase, and matrix metalloproteinase-1 (MMP-1).
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Agronomy2022, 12(10), 2426.
Environ Toxicol.2021, 36(9):1848-1856.
Inflammation.2024, 02034-1.
Elife.2021, 10:e68058.
Nutrients.2020, 12(3):595.
Eur Rev Med Pharmacol Sci.2020, 24(9):5127-5139.
Int Immunopharmacol.2021, 100:108073.
J Chromatogr B Analyt Technol Biomed Life Sci.2018, 1080:27-36
AMB Express2020. 10(1):126.
Pharmacognosy Journal, 2021, 13(5).
Related and Featured Products
J Food Prot. 2013 Sep;76(9):1539-48.
Impact of fatty acid chain length of rosmarinate esters on their antimicrobial activity against Staphylococcus carnosus LTH1502 and Escherichia coli K-12 LTH4263.[Pubmed:
23992498 ]
The effect of the addition of a newly synthesized series of rosmarinic acid (RA) estes (REs) and alcohols with chain lengths of 1, 4, 8, 10, 12, 16, and 18 carbons (RE1 to 18) on the growth behavior of Staphylococcus carnosus LTH1502 and Escherichia coli K-12 LTH4263 was investigated.
METHODS AND RESULTS:
An initial microtiter dilution assay indicated activity of compounds against S. carnosus LTH1502, whereas esters with chain lengths, RA, n-Methyl rosmarinate (RE1), n-dodecyl rosmarinate (RE12), and n-octadecyl rosmarinate (RE18) were used in a time-kill assay S. carnosus LTH1502. Compounds were added at 0.75 mM in the log phase, 5 mM in the exponential phase, 10 mM in the stationary phase. RA had no effect in the lag and exponential phase but decreased cell counts during the stationary phase. In contrast, RE1 and RE12 decreased cell number in all three phase, will RE12 reducing counts most rapidly. Addition of RE18 did not affect regardless of the growth phase.
CONCLUSIONS:
Appearance and physiological state of S. carnosus LTH1502 cells indicated difference in the way the compounds interacted with and damaged cells. Results were attributed to the different physicochemical properties of RA and its esters.
Zhongguo Zhong Yao Za Zhi. 2014 Jun;39(12):2284-8.
[Chemical constituents of Hyptis rhomboidea and their antifungal activity].[Pubmed:
25244760]
The present work is to investigate the chemical constitutions of Hyptis rhomboidea and their antifungal activities.
METHODS AND RESULTS:
The compounds were isolated by Toyopearl HW-40, Sephadex LH-20, MCI-Gel CHP-20, RP-18, PTLC and silica column chromatographic methods and subjected to evaluate some monomers antifungal activity of eight kinds of plant pathogenic bacteria. Eleven compounds were isolated and identified as ethyl caffeate (1), ursolic acid (2), oleanolic acid (3), vanillactic acid (4), Methyl rosmarinate (5), kaempferol 3-O-alpha-L-rhamnopyranosyl-(1 --> 6) -beta-D-glucopyranoside (6), kaempferol 3-O-alpha-L-rhamnopyranosyl-(1 --> 6)-beta-D-glucopyranoside (7), ilexgenin A (8), beta-amyrin (9), kaempferol 3-O-beta-D-glucopyranoside (astrgalin, 10) and cholest-5-ene-3beta, 4beta-diol (11). Compound 1 showed the strongest inhibitory effect on Sclerotinia sclerotiorum with the MIC 16.2 mg x L(-1), and compound 5 showed the strongest inhibitory effect on S. minor and Exserohilum turcicum with MIC 16.2, 8.1 mg x L(-1), respectively.
CONCLUSIONS:
All compounds were isolated from the H. rhomboidea for the first time, and compounds 1 and 5 showed antifungal activity.
Food Chem. 2011 Dec 1;129(3):884-9.
Comparative evaluation of rosmarinic acid, methyl rosmarinate and pedalitin isolated from Rabdosia serra (MAXIM.) HARA as inhibitors of tyrosinase and α-glucosidase.[Pubmed:
25212314]
Rabdosia serra has been used in traditional Chinese medicine for centuries. In order to illustrate the pharmaceutical activity of R. serra as hypoglycaemic and skin-whitening agents, rosmarinic acid (confirmed as the major compound in R. serra), Methyl rosmarinate and pedalitin isolated from R. serra were evaluated for their inhibitory effects and mechanisms on tyrosinase and α-glucosidase.
METHODS AND RESULTS:
The inhibitory effects on both tyrosinase and α-glucosidase were in decreasing order, pedalitin>Methyl rosmarinate>rosmarinic acid. The IC50 values for the tyrosinase and α-glucosidase activity inhibited by pedalitin were 0.28 and 0.29mM, respectively. Both rosmarinic acid and Methyl rosmarinate were considered as noncompetitive inhibitors of tyrosinase, while pedalitin was suggested to be a mixed-type inhibitor of tyrosinase.
CONCLUSIONS:
In the assay of α-glucosidase inhibition, rosmarinic acid was found to be a competitive inhibitor, whereas both Methyl rosmarinate and pedalitin were considered as mixed-type inhibitors.
Eur J Med Chem. 2013 Apr;62:148-57.
Synthesis of derivatives of methyl rosmarinate and their inhibitory activities against matrix metalloproteinase-1 (MMP-1).[Pubmed:
23353736]
METHODS AND RESULTS:
A series of MMP-1 inhibitors have been identified based upon a Methyl rosmarinate scaffold using structure-based drug design methods. The best compound in the series showed an IC50 value of 0.4 μM. A docking study was conducted for compound (S)-10n in order to investigate its binding interactions with MMP-1. The structure-activity relationships (SAR) were also briefly discussed.
CONCLUSIONS:
Useful SAR was established which provides important guidelines for the design of future generations of potent inhibitors against MMP-1.
Arch Pharm Res. 2004 Feb;27(2):173-6.
Antioxidative constituents from Lycopus lucidus.[Pubmed:
15022718]
Three phenolic compounds, rosmarinic acid (1), Methyl rosmarinate (2), ethyl rosmarinate (3), and two flavonoids, luteolin (4), luteolin-7-O-beta-D-glucuronide methyl ester (5) were isolated from the aerial part of Lycopus lucidus (Labiatae).
METHODS AND RESULTS:
Their structures were determined by chemical and spectral analysis. Compounds 1-5 exhibited potent antioxidative activity on the NBT superoxide scavenging assay. The IC50 values for compounds 1-5 were 2.59, 1.42, 0.78, 2.83, and 3.05 microg/mL respectively. In addition, five compounds were isolated from this plant for the first time.