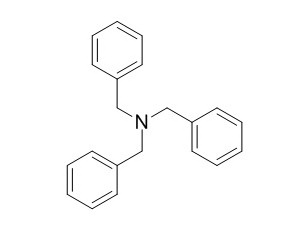
Tribenzylamine
Reference standards.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
BMC Plant Biol.2020, 20(1):214.
Food Chem.2019, 290:286-294
Bioorg Chem.2024, 150:107558.
J Pharm Pharmacol.2023, 75(9):1225-1236.
Cell Signal.2024, 124:111467.
Molecules.2017, 22(2)
Daru.2024, 32(2):689-703.
Carbohydrate Polymer Technologies & App.2021, 2:100049.
Evid Based Complement Alternat Med.2021, 2021:8850744.
J of Pharmaceutical Analysis2020, doi: 10.1016
Related and Featured Products
J Hazard Mater. 2009 Jun 15;165(1-3):886-92.
Extractive separation and determination of chromium in tannery effluents and electroplating waste water using tribenzylamine as the extractant.[Pubmed:
19135302]
A simple extractive separation method has been developed for the determination of chromium based on the extraction of Cr (VI) as its ion-pair with Tribenzylamine (TBA).
METHODS AND RESULTS:
The ion-pair is extracted at acidic pH using toluene as the diluent. The concentration of chromium in the organic phase was measured spectrophotometrically at 309 nm. The influence of experimental variables such as pH, sample volume, equilibration time, diverse ions etc. has been studied in detail. The extracted chromium (VI) could be stripped to the aqueous phase using NaOH as the stripping agent. The extracts were characterized using FT-IR spectroscopy.
A detection limit of 0.08 microg mL(-1) could be achieved and the validity of the method was checked in real tannery effluent, electroplating waste water and spiked water samples.
Inorg Chem. 2014 Apr 7;53(7):3832-8.
Tribenzylamine C-H activation and intermolecular hydrogen transfer promoted by WCl6.[Pubmed:
24624963]
METHODS AND RESULTS:
The 1:1 molar reaction of WCl6 with Tribenzylamine (tba), in dichloromethane, selectively afforded the iminium salt [(PhCH2)2N═CHPh][WCl6], 1, and the ammonium one [tbaH][WCl6], 2, in equimolar amounts. The products were fully characterized by means of spectroscopic methods, analytical methods, and X-ray diffractometry. Density functional theory calculations were carried out with the aim of comprehending the mechanistic aspects.
J Org Chem. 2011 Mar 4;76(5):1418-24.
Multiple hindered rotators in a gyroscope-inspired tribenzylamine hemicryptophane.[Pubmed:
21271707]
A gyroscope-inspired Tribenzylamine hemicryptophane provides a vehicle for exploring the structure and properties of multiple p-phenylene rotators within one molecule.
METHODS AND RESULTS:
The hemicryptophane was synthesized in three steps in good overall yield using mild conditions. Three rotator-forming linkers were cyclized to form a rigid cyclotriveratrylene (CTV) stator framework, which was then closed with an amine. The gyroscope-like molecule was characterized by (1)H NMR and (13)C NMR spectroscopy, and the structure was solved by X-ray crystallography. The rigidity of the two-component CTV-trismethylamine stator was investigated by (1)H variable-temperature (VT) NMR experiments and molecular dynamics simulations. These techniques identified gyration of the three p-phenylene rotators on the millisecond time scale at -93 °C, with more dynamic but still hindered motion at room temperature (27 °C). The activation energy for the p-phenylene rotation was determined to be ~10 kcal mol(-1). Due to the propeller arrangement of the p-phenylenes, their rotation is hindered but not strongly correlated.
CONCLUSIONS:
The compact size, simple synthetic route, and molecular motions of this gyroscope-inspired Tribenzylamine hemicryptophane make it an attractive starting point for controlling the direction and coupling of rotators within molecular systems.