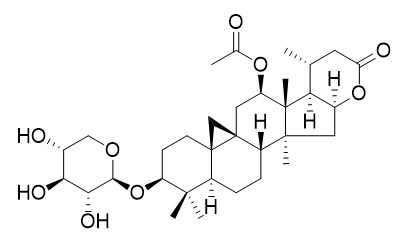
Cimilactone A
Cimilactone A shows significant anticomplement activity (IC(50)= 28.6 microm).
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Enzyme Microb Technol.2022, 153:109941.
Biology (Basel).2020, 9(11):363.
US20170000760 A12016, 42740
Toxicol Res.2019, 35(4):371-387
Phytother Res.2019, 33(5):1490-1500
Int J Mol Sci.2023, 24(23):17118.
Front Pharmacol.2021, 12:652860.
Molecules2022, 27(3),1140.
Antioxidants (Basel).2022, 11(8):1471.
J Plant Biotechnol.2023, 50:070-075.
Related and Featured Products
Beilstein J Org Chem. 2007 Jan 31;3:3.
Cimicifoetisides A and B, two cytotoxic cycloartane triterpenoid glycosides from the rhizomes of Cimicifuga foetida, inhibit proliferation of cancer cells.[Pubmed:
17266751 ]
METHODS AND RESULTS:
Two new cycloartane-type triterpene glycosides, namely cimicifoetisides A (1) and B (2), along with seven known compounds cimigenol, 25-O-acetylcimigenol, cimigenol 3-O-beta-D-xylopyranoside, 12beta-hydroxycimigenol 3-O-beta-D-xylopyranoside, cimigenol 3-O-alpha-L-arabinopyranoside, 25-deoxyshengmanol 3-O-beta-D-xylopyranoside and Cimilactone A, were isolated from the rhizomes of Cimicifuga foetida. Their structures were elucidated as cimigenol 3-O-(2'-O-acetyl)-alpha-L-arabinopyranoside (1) and 25-O-acetylcimigenol 3-O-(2'-O-acetyl)-alpha-L-arabinopyranoside (2).
CONCLUSIONS:
Both compounds 1 and 2 exhibited potent cytotoxicity against rat EAC (Ehrlich ascites carcinoma) and MDA-MB-A231 (human breast cancer) cells with IC50 values of 0.52 and 6.74 microM for 1, and 0.19 and 10.21 microM for 2, suggesting their potential for further investigation as anti-cancer agents.
Phytother Res. 2006 Nov;20(11):945-8.
Anticomplement activity of cycloartane glycosides from the rhizome of Cimicifuga foetida.[Pubmed:
16906637]
METHODS AND RESULTS:
A tetranor-cycloartane glycoside and two 9,19-cycloartane glycosides were isolated from the EtOAc-soluble fraction of the rhizome of Cimicifuga foetida. The structures of the compounds were determined to be Cimilactone A (1), 25-O-acetylcimigenol 3-O-beta-d-xylopyranoside (2) and cimigenol 3-O-alpha-l-arabinopyranoside (3), respectively, using spectroscopic analysis. The three compounds were examined for their anticomplement activity against the classical pathway of the complement system.
CONCLUSIONS:
Compound 1 showed significant anticomplement activity with an IC(50) value of 28.6 microm, whereas compounds 2 and 3 were inactive.