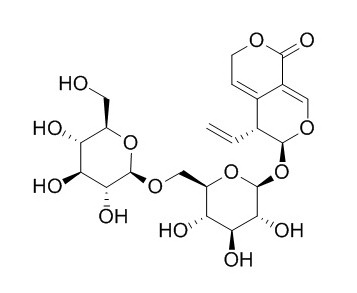
6'-O-beta-D-Glucosylgentiopicroside
6'-O-beta-D-Glucosylgentiopicroside may have antifungal activity.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
J Sep Sci.2020, 43(22):4148-4161.
FEMS Microbiol Lett.2017, 364(11)
Int J Biol Macromol.2020, 161:1230-1239.
Chinese J of Tissue Engineering Res.2022, 26(17): 2636-2641.
Molecules 2022, 27(3),1047.
Mol Cells.2015, 38(9):765-72
Metabolites.2020, 11(1):E11.
UDC.2020, 19(4).
Planta Med.2022, 88(9-10):794-804.
Drug Chem Toxicol.2024,1-12.
Related and Featured Products
Phytother Res. 2012 Feb;26(2):168-73.
Inhibitory effects of secoiridoids from the roots of Gentiana straminea on stimulus-induced superoxide generation, phosphorylation and translocation of cytosolic compounds to plasma membrane in human neutrophils.[Pubmed:
21584870]
Five secoiridoids, gentiopicroside (GTP), 6'-O-(2-hydroxy-3-O-β-D-glucopyranosyl-benzoyl)-sweroside (HGBS), 6'-O-beta-D-Glucosylgentiopicroside(GGTP), sweroside (SW) and swertiamarin (STM) were isolated from the roots of G. straminea.
METHODS AND RESULTS:
The effect of these secoiridoids on stimulus-induced superoxide generation in human neutrophils was assayed by measuring the reduction of ferricytochrome c. Tyrosyl or serine/threonine phosphorylation of neutrophil proteins, and translocation of the cytosolic compounds to the cell membrane were also investigated using specific monoclonal antibodies. The five secoiridoids used in the present experiment suppressed N-formyl-methionyl-leucyl-phenylalanine (fMLP)-induced superoxide generation in a concentration dependent manner. GTP and HGBS also suppressed phorbol 12-myristate 13-acetate (PMA) and arachidonic acid (AA)-induced superoxide generation.
However, the other three secoiridoids showed no effect on PMA- and AA-induced superoxide generation.
CONCLUSIONS:
fMLP-, PMA- and AA-induced tyrosyl or serine/threonine phosphorylation and translocation of the cytosolic proteins to the cell membrane were suppressed in parallel with the suppression of the stimulus-induced superoxide generation.
Phytochemistry, 1996, 42(5):1305-13.
Acyl secoiridoids and antifungal constituents from Gentiana macrophylla.[Pubmed:
9397205]
LC-UV-mass spectrometry and bioassay co-directed fractionation of an aqueous acetone extract of the roots of Gentiana macrophylla gave three new chromene derivatives and two novel and six known secoiridoids, along with kurarinone, kushenol I, beta-sitosterol, stigmasterol, daucosterol, beta-sitosterol-3-O-gentiobioside, alpha-amyrin, oleanolic acid, isovitexin, gentiobiose and methyl 2-hydroxy-3-(1-beta-D-glucopyranosyl)oxybenzoate.
METHODS AND RESULTS:
The structures of the new products were established from spectral and chemical evidence as 2-methoxyanofinic acid and macrophyllosides A-D. The six known secoiridoids were gentiopicroside, sweroside, 6'-O-beta-D-Glucosylgentiopicroside, 6'-O-beta-D-glucosylsweroside, trifloroside and rindoside. The new acid (2-methoxyanofinic acid), its methyl ester, kurarinone and kushenol I were shown to be active against the plant pathogenic fungus Cladosporium cucumerinum. The methyl ester and kurarinone inhibited also the growth of the human pathogenic yeast Candida albicans. Structure-activity relationships were studied. Thus, addition of a methoxyl group to the benzene nucleus of anofinic acid (2,2-dimethyl-2H-1-benzopyran-6-carboxylic acid) increased the antifungal activity remarkably whereas glycosylation at the carboxylic moiety was found to remove the activity.
CONCLUSIONS:
Esterification of the new acid induced its activity against C. albicans, but decreased its growth inhibition properties against C. cucumerinum. Hydroxylation of kurarinone at the 3 beta-position removed its activity against C. albicans and decreased the inhibition of C. cucumerinum. In addition, the chemotaxonomic significance of the identified constituents is discussed.