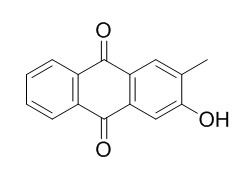
2-Hydroxy-3-methylanthraquinone
2-Hydroxy-3-methylanthraquinone enhances apoptosis of U937 cells,in part,through activation of p-p38MAPK and downregulation of p-ERK1/2; triggers caspase-3 activation mediated apoptotic induction.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Int J Mol Sci.2019, 20(14):E3538
FEBS Lett.2021, 595(20):2608-2615.
Molecules.2017, 22(12)
J.Korean Soci. Food Sci. Nutri.2024, 53(11):1166-1177
J Ethnopharmacol.2024, 320:117426.
PLoS One.2017, 12(3):e0173585
Pharmaceuticals (Basel).2024, 17(4):462.
J Ethnopharmacol.2019, 241:112025
Plant Physiol.2024, 194(4):2580-2599.
Fitoterapia.2024, 177:106138.
Related and Featured Products
J Ethnopharmacol. 2013 Jun 21;148(1):229-38.
Ethnopharmacological and bioactivity guided investigation of five TCM anticancer herbs.[Pubmed:
23623820]
Five herbs, Curcuma longa L. (CL), Scutellaria baicalensis Georgi (SBC), Scutellaria barbata D. Don (SBB), Hedyotis diffusa Willd. (HD) and Solanum nigrum L. (SN), are often prescribed in the polyherbal formulas for cancer treatment by traditional Chinese medicine (TCM) practitioners. The purpose of the present study was to identify important anticancer herbs used in TCM and carry out bioactivity-directed fractionation and isolation (BDFI) using six cancer cell lines as well as peripheral blood mononuclear cells (PBMCs), to identify constituents with anticancer activity but devoid of toxic effects against healthy immune cells.
METHODS AND RESULTS:
Of 243 document anticancer TCM treatments, 199 anticancer TCM herbs were ranked by the number of literature reports for each herb. Five herbs were identified from the top 50 ranked herbs by at least two out of three TCM practitioners as frequently used in the TCM treatment of cancer. BDFI using MTS assay was applied to determine the active anticancer extracts, fractions, and finally discrete compounds. Five herbs were selected for study of their anticancer activities. The extracts of Curcuma longa L., Scutellaria barbata D. Don, and Hedyotis diffusa showed antiproliferative activity to various extents, extracts of Scutellaria baicalensis Georgi and Solanum nigrum L. showed little anticancer activity. Seven out of the 21 fractions obtained from Hedyotis diffusa Willd. showed anticancer activity. One new compound, ethyl 13(2) (S)-hydroxy-chlorophyllide a(1), along with 10 known compounds, i.e. 2-methyl-3-methoxyanthraquinone (2), 2-hydroxymethylanthraquinone(3), 2-Hydroxy-3-methylanthraquinone(4), 2-hydroxymethy-1-hydroxyanthraquinone(5), 1-methoxy-2-hydroxyanthraquinone(6), 2-hydroxy-3-methyl-1-methoxyanthraquinone (7), oleanolic acid (8), ursolic acid (9), stigmasterol (10) and docosanoic acid (11), were isolated and identified. Compounds 2-6, 8 and 9 dose-dependently inhibited the cell viability of cancer cells within a concentration range of 1-200µM. Furthermore, compounds 2, 3, 5 and 9 showed significantly stronger inhibition of tested cancer cell lines than on that of PBMCs.
CONCLUSIONS:
This study identified anticancer herbs, extracts, fractions and eventually compounds from the documented anticancer TCM herbs by using BDFI. It also determined the antiproliferative activity in cancer and healthy immune cells of the isolated compounds from Hedyotis diffusa. The results will be useful in the validation of the clinical application of these herbs and the development of novel anticancer therapeutics.
Arch Pharm Res. 2013 Jun;36(6):752-8.
2-hydroxy-3-methylanthraquinone from Hedyotis diffusa Willd induces apoptosis in human leukemic U937 cells through modulation of MAPK pathways.[Pubmed:
23550028]
The herb of Hedyotis diffusa Willd (H. diffusa Willd), an annual herb distributed in northeastern Asia, has been known as a traditional oriental medicine for the treatment of cancer. Recently, Chinese researchers have discovered that two anthraquinones isolated from a water extract of H. diffusa Willd showed apoptosis-inducing effects against cancer cells. However, the cellular and molecular mechanisms responsible for this phenomenon are poorly understood.
CONCLUSIONS:
The current study determines the role of mitogen-activated protein kinases (MAPK) in human leukemic U937 cells apoptosis induced by 2-Hydroxy-3-methylanthraquinone from H. diffusa. Our results showed that 2-Hydroxy-3-methylanthraquinone decreased phosphorylation-ERK1/2 (p-ERK1/2), and increased p-p38MAPK, but did not affect expressions of p-JNK1/2 in U937 cells. Moreover, treatment of U937 cells with 2-Hydroxy-3-methylanthraquinone resulted in activation of caspase-3. Furthermore, PD98059 (ERK1/2 inhibitor) significantly enhanced 2-Hydroxy-3-methylanthraquinone-induced apoptosis in U937 cells, whereas caspase-3 inhibitor or SB203580 (p-p38MAPK inhibitor), decreased apoptosis in U937 cells.
CONCLUSIONS:
Taken together, our study for the first time suggests that 2-Hydroxy-3-methylanthraquinone is able to enhance apoptosis of U937 cells, at least in part, through activation of p-p38MAPK and downregulation of p-ERK1/2. Moreover, the triggering of caspase-3 activation mediated apoptotic induction.
Arch Med Res. 2011 Oct;42(7):577-83.
2-Hydroxy-3-methylanthraquinone from Hedyotis diffusa WILLD induces apoptosis via alteration of Fas/FasL and activation of caspase-8 in human leukemic THP-1 cells.[Pubmed:
22108623]
Numerous studies have shown that Hedyotis diffusa WILLD (H. diffusa) could promote apoptosis in several cancer cell lines. However, the cellular and molecular mechanisms responsible for this phenomenon are still poorly understood. The current study determines the role of Fas/FasL in THP-1 cell apoptosis induced by 2-Hydroxy-3-methylanthraquinone from H. diffusa.
METHODS AND RESULTS:
THP-1 cells were treated with 2-Hydroxy-3-methylanthraquinone from H. diffusa. The effect on the cell viability of THP-1 cells was evaluated using the trypan blue assay, and cell apoptosis was measured by Giemsa staining and flow cytometry. Protein expression was assayed using the Western blot method. Our results showed that 2-Hydroxy-3-methylanthraquinone from H. diffusa induced THP-1 cell apoptosis in a time- and dose-dependent manner. Apoptosis was associated with a more prominent induction expression of Fas/FasL, DR4 and TRAIL in a time-dependent manner. Moreover, treatment of THP-1 cells with 2-Hydroxy-3-methylanthraquinone from H. diffusa resulted in activation of caspase-8.
CONCLUSIONS:
For the first time, our study suggests that 2-Hydroxy-3-methylanthraquinone from H. diffusa is able to enhance apoptosis of THP-1 cells, at least in part, through activation of Fas/FasL, DR4 and TRAIL. Moreover, triggering of caspase-8 activation mediated apoptotic induction.
Zhong Yao Cai. 2014 Apr;37(4):602-4.
[Chemical constituents of Juncus setchuensis].[Pubmed:
25345132]
To study the chemical constituents of Juncus setchuensis.
METHODS AND RESULTS:
Column chromatography was used in the isolation procedure. The structures of isolated compounds were elucidated by spectral data. Eight compounds were isolated and their structures were identified as 2-Hydroxy-3-methylanthraquinone (1), physcion (2), stigmasterol (3), stigmast-3,6-dione (4), vanillin (5), n-heptacosanoic acid (6), trans-hydroxycinnamic (7) and 4-hydroxy-3-methoxy benzoic acid (8).
CONCLUSIONS:
Compound 1, 2, 4 and 6 are obtained from this genus for the first time and all the compounds are obtained from this plant for the first time.