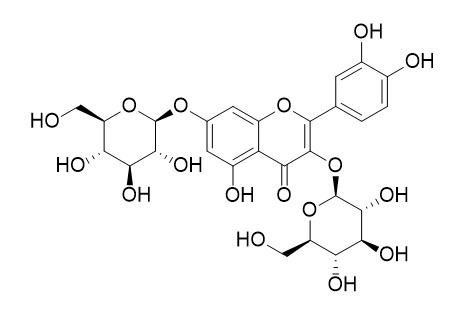
Quercetin 3,7-diglucoside
Quercetin 3,7-diglucoside exhibits antioxidant activity.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Cells.2022, 11(8), 1311.
Food Res Int.2024, 197(Pt 1):115244.
Plant Physiol.2024, 194(4):2580-2599.
Kor. J. Herbol.2019, 34(2):59-66
Inflammation.2015, 38(4):1502-16
Pharmaceuticals (Basel).2024, 17(8):988.
Reprod Toxicol.2020, 96:1-10.
Agronomy2020, 10(10),1489
Cosmetics2025, 12(3), 108
Int J Mol Sci.2021, 22(16):8641.
Related and Featured Products
Physiol Mol Biol Plants . 2018 Sep;24(5):881-887.
Impact of maturity on phenolic composition and antioxidant activity of medicinally important leaves of Ficus carica L[Pubmed:
30150862]
Ficus carica L. is one of the oldest trees belonging to the mulberry family. It has both nutritional and medicinal benefits, as it is a source of potential bioactive compounds. Fig leaves were collected at four maturation stages, to evaluate the variation in polyphenolic compounds, antioxidant activity, and different pigments level. Polyphenols were identified using HPLC-DAD. Different pigments level, total phenolic contents, and radical scavenging activity were quantified. The HPLC analysis revealed the presence of fourteen polyphenolic compounds in the fig leaves. Quercetin-3-glucoside, caftaric acid, quercetin-3, 7-diglucoside, and coumaroyl-hexose were the major phenolic compounds. Kaempferol-3-O-sophorotrioside, cichoric acid and sinapic acid glucoside were identified for the first time. Significant quantitative changes were observed during maturation, particularly in polyphenolic compounds. There were significant changes in pigments level, total phenolic contents, and radical scavenging activity. It was observed that fig leaves are a rich source of polyphenolic compounds during all stages of maturation.
J Sci Food Agric . 2019 Feb;99(3):1046-1054.
Preliminary evaluation of quince (Cydonia oblonga Mill.) fruit as extraction source of antioxidant phytoconstituents for nutraceutical and functional food applications[Pubmed:
30014572]
Background: Quince (Cydonia oblonga) fruits can be considered as starting material for the extraction of health-promoting phytochemicals, to be exploited in food and nutraceuticals. In the present work, liquid chromatography coupled with diode array detection and tandem mass spectrometry analysis allowed the study of the phytochemical composition of quince fruits and to compare it with those of six commercial apple varieties.
Results: The distribution and quantification of secondary metabolites in peel and pulp were studied and compared with six commercial apple varieties. Furthermore the in vitro antioxidant activity was determined by 2,2-diphenyl-2-picrylhydrazyl (DPPH) assay. Quince fruit presented significant amounts of shikimic and quinic acid derivatives, as well as flavonoids and procyanidins. Compared with apple, quince fruit composition was characterized by the presence of 4-caffeoylshikimic acid, 4-caffeoyl quinic acid, quercetin-3,7-diglucoside, kaempferol-3-O-rhamnoside and kaempferol-7-O-glucoside, and the dihydrochalcones were not detectable. The peel showed the highest contents of phenolics, whereas 3-O-caffeoylquinic acid was the most abundant compound in the quince pulp. The Pearson correlation index was calculated considering the quantitative amount of the phenolic constituents and the radical scavenging activity toward DPPH both for peel and pulp extracts. ·
Conclusions: This study highlighted the presence of significant amounts of valuable secondary metabolites in quince fruit, in particular the procyanidins and caffeoyl esters with shikimic and quinic acid. Notably, owing to the higher content in phenolic compounds and the stronger antioxidant capacity compared with the other fruits considered, the use of C. oblonga as a source of antioxidant can be valuable in nutraceuticals, revealing new possible applications of quince fruit. © 2018 Society of Chemical Industry.
Food Funct . 2017 Jan 25;8(1):429-436.
Protective effects of dietary glycine and glutamic acid toward the toxic effects of oxidized mustard oil in rabbits[Pubmed:
28091680]
The protective role of glycine and glutamic acid against the toxic effects of oxidized oil was studied for the first time. Mustard seed oil was thermally oxidized and characterized for quality characteristics and polyphenolic composition using reversed phase HPLC-DAD. Significant changes in the quality characteristics occurred with thermal oxidation. Fourteen polyphenolic compounds were identified and quantified in oils. Quercetin-3-glucoside, quercetin-3-feruloylsophoroside, catechin, quercetin-3-rutinoside, quercetin-3,7-diglucoside, sinapic acid and vanillic acid hexoside were the major compounds in the fresh and oxidized oil. Oxidized, un-oxidized mustard oils, glycine and glutamic acid were given to rabbits alone or in combination. The biochemical responses were studied in terms of haematological and biochemical parameters and histopathology. It has been observed that biochemical and haematological parameters were adversely affected by the oxidized oil, while supplementation of both amino acids was beneficial in normalizing these parameters. Both amino acids alone have no significant effects, however, oxidized oil affected the liver by enhancing fat accumulation, causing hepatitis, reactive Kupffer cells and necrosis. The co-administration of oxidized oils with glycine or glutamic acid revealed significant recovery of the liver structure and function. In conclusion, glycine or glutamic acid is beneficial and protective against food toxicity and can be considered as an ameliorative food supplement.
J Chem Ecol . 1987 Mar;13(3):473-493.
Flavonoid pigments in chalkhill blue (Lysandra coridon Poda) and other lycaenid butterflies[Pubmed:
24301889]
Nine flavonoids, namely, kaempferol, kaempferol 7-rharanoside, kaempferol 3-rhamnoside, kaempferol 3-glucoside, kaempferol 3-glucoside, 7-rhamnoside, quercetin 3-glucoside, Quercetin 3,7-diglucoside, isorhamnetin 3-glucoside, and isorhamnetin 3,7-diglucoside, have been identified in the body and wings of the chalkhill blue butterflyLysandra coridon Poda. Flavonoids have also been found in 15 of a further 17 lycaenid species examined. Analysis of the two-dimensional paper chromatographic flavonoid patterns and aglycone results has shown that the flavonoid content ofL. coridon and the other lycaenids is dependent on the flavonoid content of the larval diet. Differences in the flavonoid patterns ofL. coridon and its leguminous larval food plantsHippocrepis comosa, Anthyllis vulneraria, andLotus corniculatus, indicate that the ingested flavonoids are metabolized byL. coridon or its gut flora before sequestration. Despite the presence of fiavones, glycoflavones, and isoflavones in the larval food plant species, only flavonols are sequestered by the lycaenid species examined. The relationship between lycaenid butterflies and their larval food plants, and the possible role(s) of flavonoids in lycaenids has been discussed. Interactions between ants, plants, flavonoids, and myrmecophilous lycaenids have also been considered.
Nat Prod Res . 2014;28(6):377-382.
Anti-influenza A virus activity of a new dihydrochalcone diglycoside isolated from the Egyptian seagrass Thalassodendron ciliatum (Forsk.) den Hartog[Pubmed:
24443884]
One new dihydrochalcone diglycoside has been isolated from the EtOAc fraction of the Egyptian seagrass Thalassodendrin ciliatum (Forsk.) den Hartog, and was identified as 6'-O-rhamnosyl-(1‴ → 6″)-glucopyranosyl asebogenin for which a trivial name Thalassodendrone was established. Furthermore, five known phenolics were isolated and identified as asebotin, Quercetin 3,7-diglucoside, protocatechuic acid, ferulic acid and p-hydroxybenzoic acid. The structures of all the isolated compounds were established based on 1D and 2D NMR spectroscopy and high-resolution-mass spectrometer. High-resolution electrospray ionization mass spectra (HR-ESI-MS) were obtained using a JEOL JMS-T100TD spectrometer (JEOL Ltd., Tokyo, Japan). The anti-influenza A virus activity of the isolated new compound and asebotin was evaluated, and the obtained results revealed that the inhibition dose concentration of asebotin was more than that of Thalassodendrone with IC50 = 2.00 and 1.96 μg/mL, respectively, and with cytotoxic concentration (CC50) of 3.36 and 3.14 μg/mL, respectively.