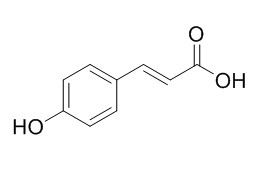
p-Hydroxy-cinnamic acid
p-Hydroxy-cinnamic acid may have in vitro antimalarial activity. p-Hydroxycinnamic acid and β-cryptoxanthin have anti-osteoporotic properties in vivo, they can antagonize NF-κB activation in MC3T3 preosteoblastic cells。
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Mie University2019, 10076.
Institute of Food Science & Technology2021, 56(11).
Biol Pharm Bull.2018, 41(1):65-72
J Food Drug Anal.2023, 31(2):254-277.
Asian J Beauty Cosmetol2024, 22(1): 103-112.
Front Plant Sci.2022, 12:811166.
Phytomedicine.2015, 22(11):1027-36
Oncol Rep.2021, 46(1):143.
J Biomed Sci.2020, 27(1):60.
J Ethnopharmacol.2017, 209:305-316
Related and Featured Products
Parasitol Res. 2012 Feb;110(2):539-44.
Antimalarial drug interactions of compounds isolated from Kigelia africana (Bignoniaceae) and their synergism with artemether, against the multidrug-resistant W2mef Plasmodium falciparum strain.[Pubmed:
21814840]
For decades, drug resistance has been the major obstacle in the fight against malaria, and the search for new drugs together with the combination therapy constitutes the major approach in responding to this situation.
METHODS AND RESULTS:
The present study aims at assessing the in vitro antimalarial activity of four compounds isolated from Kigelia africana stem bark (atranorin - KAE1, specicoside - KAE7, 2β,3β,19α-trihydroxy-urs-12-20-en-28-oic acid - KAE3, and p-Hydroxy-cinnamic acid - KAE10) and their drug interactions among themselves and their combination effects with quinine and artemether. The antiplasmodial activity and drug interactions were evaluated against the multidrug-resistant W2mef strain of Plasmodium falciparum using the parasite lactate dehydrogenase assay. Three of the four compounds tested were significantly active against W2mef: specicoside (IC(50) = 1.02 ± 0.17 μM), 2β,3β,19α-trihydroxy-urs-12-en-28-oic acid (IC(50) = 1.86 ± 0.15 μM) and atranorin (IC(50) = 1.78 ± 0.18 μM), whereas p-Hydroxy-cinnamic acid showed a weak activity (IC(50) = 12.89 ± 0.87 μM). A slight synergistic effect was observed between atranorin and 2β,3β,19α-trihydroxy-urs-12-en-28-oic acid (Combination index, CI = 0.82) whereas the interaction between specicoside and p-Hydroxy-cinnamic acid were instead antagonistic (CI = 2.67).
CONCLUSIONS:
All the three compounds showed synergistic effects with artemether, unlike the slight antagonistic interactions of atranorin and 2β,3β,19α-trihydroxy-urs-12-en-28-oic acid in combination with quinine. K. africana compounds are therefore likely to serve as leads in the development of new partner drugs in artemether-based combination therapy.
Nat Prod Commun. 2012 Oct;7(10):1351-2.
Thymofolinoates A and B, new cinnamic acid derivatives from Euphorbia thymifolia.[Pubmed:
23157007]
Two new cinnamic acid derivatives, thymofolinoates A (1) and B (2) have been isolated from the chloroform soluble fraction of Euphorbia thymifolia and their structures assigned from 1H and 13C NMR spectra, DEPT and by 2 D COSY, HMQCand H MBC experiments. In addition, p-Hydroxy-cinnamic acid(3), 5-hydroxy-6,7,8,4'-tetramethoxy flavone (4), and 5-hydroxy-3',4',6,7,8-pentamethoxy flavone (5) have also been isolated for the first time from this species.
Mol Med Rep. 2009 Jul-Aug;2(4):641-4.
The bone anabolic carotenoids p-hydroxycinnamic acid and β-cryptoxanthin antagonize NF-κB activation in MC3T3 preosteoblasts.[Pubmed:
21475879]
The carotene p-hydroxycinnamic acid and the xanthophyll β-cryptoxanthin are members of the carotenoid family of plant-derived pigments, which are endowed with anti-osteoporotic properties in vivo. p-Hydroxycinnamic acid and β-cryptoxanthin have been demonstrated to stimulate osteoblastic bone formation while simultaneously repressing osteoclastic bone resorption in vitro. However, their mechanisms of action remain poorly elucidated. It is well established that the NF-;kgr;B signal transduction pathway plays a critical role in osteoclast differentiation. Moreover, we recently demonstrated that NF-κB activity potently antagonizes osteoblastic differentiation and mineralization in vitro.
METHODS AND RESULTS:
In this study, we used transient transfection assays of a NF-κB luciferase reporter to demonstrate that p-hydroxycinnamic acid and β-cryptoxanthin antagonize NF-κB activation in MC3T3 preosteoblastic cells.
CONCLUSIONS:
The data obtained suggest that NF-κB may be a common molecular target by which several bone active agents, including carotenoids, promote osteoblastic bone formation.
Yao Xue Xue Bao. 2014 Aug;49(8):1150-4.
Chemical constituents of lateral roots of Aconitum carmichaelii Debx.[Pubmed:
25322557]
In order to find the cardiotonic constituents of lateral roots of Aconitum carmichaelii Debx., the investigation was carried out.
METHODS AND RESULTS:
Silica gel column chromatography, Sephadex LH-20, medium-pressure MCI and reverse phase ODS column chromatography were used to separate the 90% EtOH extract of the lateral roots of Aconitum carmichaelii Debx. The structures of the isolated compounds have been identified by chemical properties and spectroscopic analyses. Ten compounds were isolated and their structures were elucidated as benzoic acid-5-hydroxy-2-benzoyl-amino methyl ester (1), honokiol (2), pinoresinol (3), salicylic acid (4), p-Hydroxy-cinnamic acid (5), songorine (6), karakoline (7), mesaconitine (8), hypaconitine (9) and 14-benzoylhypaconitine (10), separetely. Compound 1 is a new compound and its structure has been established by NMR, HR-ESI-MS, UV, IR and X-Ray.
CONCLUSIONS:
Compound 2-5 are isolated from the lateral roots of Aconitum carmichaelii Debx. for the first time.