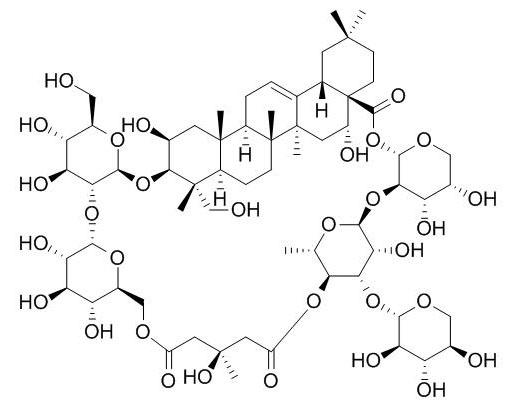
Tubeimoside III
Tubeimoside III has anti-inflammatory, anti-tumor, and anti-tumorigenic activities, stronger than those of tubeimoside II. It has acute toxicity, stronger than that of tubeimoside II.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Front Nutr.2024, 11:1409309.
Molecules.2024, 29(5):1050.
J Ethnopharmacol.2022, 289:115018.
Molecules.2016, 21(6)
J. Food Composition and Analysis2022, 114:104731
Pharmaceuticals (Basel).2024, 17(3):352.
Int J Mol Sci.2020, 21(19):7209.
BMC Complement Altern Med.2018, 18(1):221
Nutrients.2024, 16(15):2518.
Aging (Albany NY).2023, 15(24):15557-15577.
Related and Featured Products
Acta Pharmacol Sin. 2001 May;22(5):463-8.
Structure-activity relationship of tubeimosides in anti-inflammatory, antitumor, and antitumor-promoting effects.[Pubmed:
11743898]
To study structure-activity relationship of tubeimosides isolated from Bolbostemma paniculatum for their anti-inflammatory, antitumor, and antitumor-promoting effects.
METHODS AND RESULTS:
Tubeimoside I, Tubeimoside II, and Tubeimoside III were isolated from tubers of Bolbostemma paniculatum (Maxim) Franquet (Cucurbitaceae), a Chinese folk medicine,"Tubeimu", and their anti-inflammatory, anti-tumor, anti-tumorigenic activities, and acute toxicity were studied in vivo.
Tubeimoside I, Tubeimoside II, and Tubeimoside III are all natural analogues of oleanane type of triterpenoid saponins from the same medicinal plant, and all show anti-inflammatory, antitumor, and antitumor-promo ting effects. However, the anti-inflammatory, anti-tumor, and anti-tumorigenic activities of Tubeimoside II are stronger than those of tubeimoside I, and the acute toxicity of Tubeimoside II is lower than that of tubeimoside I; the anti-inflammatory, anti-tumor, and anti-tumorigenic activities of Tubeimoside III are stronger than those of Tubeimoside II, and the acute toxicity of Tubeimoside III is also stronger than that of Tubeimoside II.
CONCLUSIONS:
C-16 hydroxyl group of Tubeimoside II plays an important role in enhancing biological activity of Tubeimoside II and in decreasing its toxicity. The difference of chemical structure in B and/or C position between tubeimosides III and II plays an important role in enhancing biological activity and toxicity of Tubeimoside III. Therefore tubeimosidre II may be the most promising agent for cancer chemoprevention and chemotherapy among tubeimosides I, II, and III.
Carcinogenesis. 1995 Dec;16(12):3045-8.
Inhibition of the tumor promoting action of 12-O-tetradecanoylphorbol-13-acetate by tubeimoside III isolated from Bolbostemma paniculatum.[Pubmed:
8603483]
METHODS AND RESULTS:
As tubeimoside I isolated from Bolbostemma paniculatum (Maxim.) Franquet (Cucurbitaceae) has been shown to suppress tumor promoter effects, Tubeimoside III from the same plant was tested in vitro and in vivo against the action of the tumor promoter 12-O-tetradecanoylphorbol-13-acetate (TPA). Tubeimoside III, the natural analog of tubeimoside I, also had an anti-inflammatory effect on mouse ear edema induced by arachidonic acid and TPA and a potent anti-tumor promoting effect on two-stage carcinogenesis of mouse skin after topical application. However, the important difference in bioactivities between tubeimosides I and III is the non-activity of Tubeimoside III as an inhibitor of tumor promotion if administered orally.
CONCLUSIONS:
Differences in metabolism connected with different routes of the compound may be one of a number of explanations of the important difference.