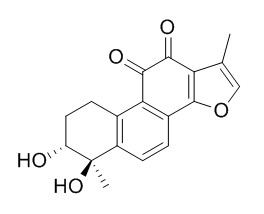
Tanshindiol C
Tanshindiol C protects macrophages from oxLDL induced foam cell formation via activation of Prdx1/ABCA1 signaling and that Prdx1 may be a novel target for therapeutic intervention of atherosclerosis.It possesses a unique anti-cancer activity whose mechanism involves the inhibition of EZH2 activity and would provide chemically valuable information for designing a new class of potent EZH2 inhibitors.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
An Acad Bras Cienc.2023, 95(3):e20220672
Natural Product Communications2020, doi: 10.1177.
Nutrients.2023, 15(13):2960.
Int J Mol Sci.2018, 19(9):E2825
Journal of Apicultural Research2021, 60(1)
Metabolites. 2023, 13(11):1122.
Virol J.2024, 21(1):95.
Research SPJ.2024, 0377.
Curr Issues Mol Biol.2023, 45(3):2136-2156.
Hum Exp Toxicol.2023, 42:9603271231171642.
Related and Featured Products
Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease, 2017, 1864(3):882-890.
Tanshindiol C inhibits oxidized low-density lipoprotein induced macrophage foam cell formation via peroxiredoxin 1 dependent pathway.[Reference:
WebLink]
NF-E2-related factor 2 (Nrf2) has been shown to be protective in atherosclerosis. The loss of Nrf2 in macrophages enhances foam cell formation and promotes early atherogenesis. Tanshindiol C (Tan C) is isolated from the root of Salvia miltiorrhiza Bge., a traditional Chinese medicine that has been used for the treatment of several cardiovascular diseases for many years. This study was aimed to test the potential role of Tan C against macrophage foam cell formation and to explore the underlying mechanism.
METHODS AND RESULTS:
Firstly, we observed that Tan C markedly suppressed oxidized low-density lipoprotein (oxLDL) induced macrophage foam cell formation. Then, we found that Tan C was an activator of both Nrf2 and Sirtuin 1 (Sirt1) in macrophages. Nrf2 and Sirt1 synergistically activated the transcription of anti-oxidant peroxiredoxin 1 (Prdx1) after Tan C treatment. More important, we demonstrated that silencing of Prdx1 promoted oxLDL-induced macrophage foam cell formation. Prdx1 upregulated adenosine triphosphate-binding cassette (ABC) transporter A1 (ABCA1) expression and decreased intracellular lipid accumulation. Furthermore, Tan C ameliorated oxLDL induced macrophage foam cell formation in a Prdx1-dependent manner.
CONCLUSIONS:
These observations suggest that Tan C protects macrophages from oxLDL induced foam cell formation via activation of Prdx1/ABCA1 signaling and that Prdx1 may be a novel target for therapeutic intervention of atherosclerosis.
Bioorganic & Medicinal Chemistry Letters, 2014, 24(11):2486-2492.
Biological evaluation of tanshindiols as EZH2 histone methyltransferase inhibitors.[Reference:
WebLink]
EZH2 is the core subunit of Polycomb repressive complex 2 catalyzing the methylation of histone H3 lysine-27 and closely involved in tumorigenesis.
METHODS AND RESULTS:
To discover small molecule inhibitors for EZH2 methyltransferase activity, we performed an inhibitor screen with catalytically active EZH2 protein complex and identified tanshindiols as EZH2 inhibitors. Tanshindiol B and Tanshindiol C potently inhibited the methyltransferase activity in in vitro enzymatic assay with IC50 values of 0.52 μM and 0.55 μM, respectively. Tanshindiol C exhibited growth inhibition of several cancer cells including Pfeiffer cell line, a diffuse large B cell lymphoma harboring EZH2 A677G activating mutation. Tanshindiol treatment in Pfeiffer cells significantly decreased the tri-methylated form of histone H3 lysine-27, a substrate of EZH2, as revealed by Western blot analysis and histone methylation ELISA. Based on enzyme kinetics and docking studies, we propose that tanshindiol-mediated inhibition of EZH2 activity is competitive for the substrate S-adenosylmethionine.
CONCLUSIONS:
Taken together, our findings strongly suggest that tanshindiols possess a unique anti-cancer activity whose mechanism involves the inhibition of EZH2 activity and would provide chemically valuable information for designing a new class of potent EZH2 inhibitors.
Phytochemistry, 1985, 24(4):815-817.
Pigments from Salvia miltiorrhiza.[Reference:
WebLink]
METHODS AND RESULTS:
Five new o-naphthaquinone diterpenes, tanshindiol A, tanshindiol B, Tanshindiol C, nortanshinone and 3α-hydroxytanshinone IIA, have been isolated from the roots of Salvia miltiorrhiza as minor components. Their relative stereochemistries have been established on the basis of spectral and chemical evidence.