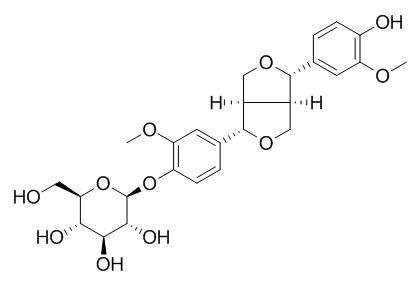
Pinoresinol 4-O-beta-D-glucopyranoside
(+)-Pinoresinol 4-O-beta-D-glucopyranoside revealed that the presence of a vanilloyl group in the sugar moiety of (+)-pinoresinol 4-O-[6″-O-vanilloyl]-β-D-glucopyranoside is crucial for its anti-influenza virus activity.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Viruses.2024, 16(7):1128.
J Biomed Sci.2020, 27(1):60.
Appl. Sci.2023, 13(17):9984.
J Separation Science & Technology2016, 51:1579-1588
Enzyme Microb Technol.2019, 122:64-73
Pol J Microbiol.2021, 70(1):117-130.
Evid Based Complement Alternat Med.2016, 2016:1739760
Food Funct.2020, 11(2):1322-1333.
Plant Physiol.2023, 193(3):1758-1771.
Molecules.2022, 27(22):7887.
Related and Featured Products
PLoS One. 2014 Aug 7;9(8):e104544.
In vitro anti-influenza virus activities of a new lignan glycoside from the latex of Calotropis gigantea.[Pubmed:
25102000]
A new lignan glycoside, (+)-pinoresinol 4-O-[6″-O-vanilloyl]-β-D-glucopyranoside (1) and two known phenolic compounds, 6'-O-vanilloyltachioside (2) and 6'-O-vanilloylisotachioside (3) were isolated from the latex of Calotropis gigantea (Asclepiadaceae).
METHODS AND RESULTS:
The structure of the new compound was elucidated by using spectroscopic and chemical methods. Three isolates (1-3) and one authentic compound, (+)-
Pinoresinol 4-O-beta-D-glucopyranoside, were screened for A/PR/8/34 (H1N1) inhibitory activity by cytopathic effect (CPE) inhibition assay on MDCK cells. Compound 1 showed inhibitory activity against A/PR/8/34 (H1N1). In sharp contrast, the other three compounds (2, 3 and (+)-Pinoresinol 4-O-beta-D-glucopyranoside) did not show such activity. An analysis of structure-activity relationship between 1 and
(+)-Pinoresinol 4-O-beta-D-glucopyranoside revealed that the presence of a vanilloyl group in the sugar moiety of 1 is crucial for its anti-influenza virus activity. Compound 1 was further evaluated for in vitro inhibitory activities against a panel of human and avian influenza viruses by CPE inhibition assay. It showed inhibitory effect against human influenza viruses in both subtypes A and B (IC50 values around 13.4-39.8 µM with SI values of 3.7-11.4), while had no effect on avian influenza viruses. Its antiviral activity against human influenza viruses subtype A was further confirmed by plaque reduction assay. The time course assay indicated that 1 exerts its antiviral activity at the early stage of viral replication. A mechanistic study showed that 1 efficiently inhibited influenza virus-induced activation of NF-κB pathway in a dose-dependent manner, but had no effect on virus-induced activation of Raf/MEK/ERK pathway. Further studies demonstrated that nuclear translocation of transcription factor NF-κB induced by influenza virus was significantly blocked by 1, meanwhile, nuclear export of viral ribonucleoproteins was also effectively inhibited.
CONCLUSIONS:
These findings suggest that this new lignan glycoside from Calotropis gigantea, may have therapeutic potential in influenza virus infection through inhibition of NF-κB pathway and viral ribonucleoproteins nuclear export.