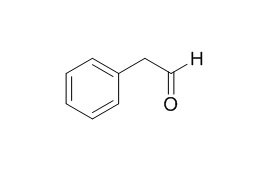
Phenylacetaldehyde
Phenylacetaldehyde is a oviposition inhibitor for the Pink Bollworm, it can attract Moths to bladder flower and to blacklight traps. Phenylacetaldehyde and carbon monoxide as effective additives for the selective hydrogenation of cyclooctadienes to cyclooctene over palladium catalysts.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Int J Anal Chem.2017, 2017:1254721
Antioxidants (Basel).2021, 10(11): 1802.
Indian J. of Experimental Bio.2020, 9(58).
ACS Omega.2021, 6(36):23460-23474.
Asian J Beauty Cosmetol2021, 19(1): 57-64.
Phytomedicine.2019, 59:152785
BMC Complement Altern Med.2019, 19(1):367
J Agric Food Chem.2021, 69(14):4210-4222.
Psychopharmacology (Berl).2020, 10.1007
LWT-Food Sci Technol2020, 109163
Related and Featured Products
Environmental Entomology, 1979 ,8 (3) :444-7.
Phenylacetaldehyde Attracts Moths to Bladder Flower and to Blacklight Traps[Reference:
WebLink]
The fragrant chemical compound volatilized by the bladder flower, Araujia sericofera Brot., was identified as Phenylacetaldehyde. It attracted many economically important species of moths, and when it was used with a blacklight trap, it increased trap collections of pest moth species by 48%.
Journal of Economic Entomology , 1977 , 70 (5) :547-8.
Phenylacetaldehyde: Oviposition Inhibitor for the Pink Bollworm[Reference:
WebLink]
Field plots of cotton treated with methyl salicylate or untreated had significantly greater infestations of Pectinophora gossypiella (Saunders) than similar plots treated with Phenylacetaldehyde.
METHODS AND RESULTS:
In laboratory tests, Phenylacetaldehyde did not reduce hatch when applied to eggs of the pink bollworm oviposited on cotton leaves. However, laboratory female moths laid ca. 75% of their eggs on untreated plants when caged with treated and untreated cotton plants.
CONCLUSIONS:
Phenylacetaldehyde appears to be an oviposition inhibitor for the pink bollworm and may be useful for control.
Bulletin of the Chemical Society of Japan, 2006 , 65 (11) :2960-5.
Phenylacetaldehyde and Carbon Monoxide as Effective Additives for the Selective Hydrogenation of Cyclooctadienes to Cyclooctene over Palladium Catalysts[Reference:
WebLink]
METHODS AND RESULTS:
Three isomeric cyclooctadienes (1,5-, 1,4-, and 1,3-COD) were hydrogenated with Phenylacetaldehyde (PAA)- or carbon monoxide (CO)-treated palladium black in tetrahydrofuran at 25-degrees-C and atmospheric pressure of hydrogen. A complete depression of the hydrogenation of cyclooctene (COE) to cyclooctane (COA) was attained with both of the poisoned catalysts. By PAA the hydrogenation of 1,5-COD to COE was not hindered and the isomerization of 1,5-COD to 1,4-COD was greatly promoted, while the rate of hydrogenation of 1,4- and 1,3-COD decreased in the presence of PAA. By CO the hydrogenation and isomerization of 1,5-COD, as well as the hydrogenation of 1,4- and 1,3-COD, was suppressed. In the hydrogenation of 1,5-COD the PAA-treated palladium was more active and selective than was the CO-treated palladium for the formation of COE.
CONCLUSIONS:
The differences in the effects of these catalyst poisons are discussed on the basis of kinetic data concerning the individual and competitive hydrogenation of the three isomeric cyclooctadienes.