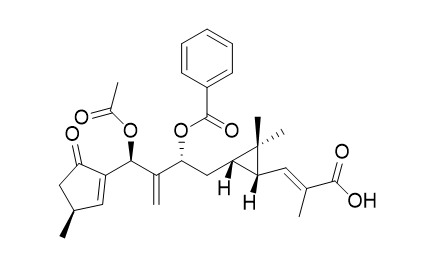
Lathyranoic acid A
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Front Immunol. 2020, 11:62.
Biomolecules.2024, 14(10):1257.
Korean J. Medicinal Crop Sci.2021, 29(6):425-433
Korean J Acupunct2020, 37:104-121
Front Pharmacol.2021, 12:744624.
Asian J Beauty Cosmetol2016, 14(3):249-257
Biomed Pharmacother.2022, 145:112474.
Food Quality and Safety2018, 2:213-219
J Biotechnol.2020, 318:10-19.
J Cell Mol Med.2024, 28(16):e70016.
Related and Featured Products
J Org Chem . 2011 Mar 4;76(5):1448-51.
Total synthesis of lathyranoic acid A[Pubmed:
21275399]
The first total synthesis of Lathyranoic acid A (1) was accomplished stereoselectively in a linear sequence of 20 steps and an overall yield of 1.4%. This modular synthesis featured a cyclic, stereocontrolled Cu-catalyzed intramolecular cyclopropanation to construct the cis-cyclopropane unit, a Grubbs metathesis to construct the γ-substituted cyclopentenone moiety, and an anion-mediated conjugate addition.
Org Lett . 2005 Mar 31;7(7):1379-82.
Lathyranoic acid A: first secolathyrane diterpenoid in nature from Euphorbia lathyris[Pubmed:
15787511]
Lathyranoic acid A (1), the first secolathyrane diterpenoid with an unprecedented skeleton, and a new diterpenoid Euphorbia factor L11 (2) were isolated from the seeds of Euphorbia lathyris. Their structures were elucidated by spectroscopic analysis and chemical methods. A biogenetic route involving an enzymatic Baeyer-Villiger oxidation as the key step was postulated for the transformation of 2 to 1 and mimicked by an unusual chemical Baeyer-Villiger oxidation.