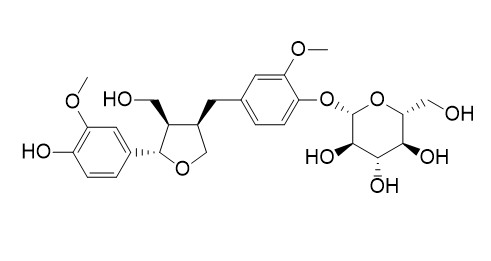
Lariciresinol 4-O-glucoside
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Horticulturae2024, 10(4), 382.
Antioxidants (Basel).2020, 9(2): E119
Preprints2022, 2022030063.
Plant Physiol Biochem.2019, 144:355-364
Food Funct.2023, 14(9):4354-4367.
Int J Mol Sci.2024, 25(22):12152.
Toxicol In Vitro.2019, 59:161-178
J Appl Microbiol.2024, 135(7):lxae180.
Korean Journal of Pharmacognosy2017, 48(4):320-328
Phytomedicine.2019, 58:152893
Related and Featured Products
J Agric Food Chem . 2001 Jun;49(6):2788-2798.
Isolation and characterization of novel benzoates, cinnamates, flavonoids, and lignans from Riesling wine and screening for antioxidant activity[Pubmed:
11409967]
A German Riesling wine has been fractionated with the aid of countercurrent chromatography. After purification by HPLC, the structures of 101 compounds were established by mass spectrometry and NMR spectroscopy. Seventy-three of the isolated compounds exhibited a phenolic or benzylic structure. Fifty-four compounds were reported for the first time as Riesling wine constituents. New compounds identified in this work included twelve benzoic and cinnamic acid derivatives. In addition to two isomeric (E)-caffeoyl ethyl tartrates, the glucose esters of (E)-cinnamic, (E)-p-coumaric, and (E)-ferulic acid, as well as the 4-O-glucosides of (E)- and (Z)-ferulic acid, have been identified for the first time in Riesling wine. The structures of two additional phenylpropanoids were elucidated as 3-hydroxy-1-(4-hydroxy-3-methoxyphenyl)-propan-1-one and 2,3-dihydroxy-1-(4-hydroxy-3-methoxyphenyl)-propan-1-one. Moreover, two ethyl esters, i.e., ethyl protocatechuate and ethyl gallate, as well as the glucose ester of vanillic acid, were newly detected in Riesling wine. Novel representatives in the flavonoid group were dihydrokaempferol, dihydroquercetin, and four dihydroflavonol glycoconjugates, i.e., the 3-O-glucosides of dihydrokaempferol and dihydroquercetin, as well as the 3-O-xyloside and the 3'-O-glucoside of dihydroquercetin. Additionally, six novel lignans, i.e., Lariciresinol 4-O-glucoside, three isolariciresinol derivatives, and two secoisolariciresinols, as well as three neolignans were isolated. Structural elucidation of the newly isolated wine constituents is reported together with the determination of their antioxidant activity.
J Agric Food Chem . 2001 Jun;49(6):2788-2798.
Isolation and characterization of novel benzoates, cinnamates, flavonoids, and lignans from Riesling wine and screening for antioxidant activity[Pubmed:
11409967]
A German Riesling wine has been fractionated with the aid of countercurrent chromatography. After purification by HPLC, the structures of 101 compounds were established by mass spectrometry and NMR spectroscopy. Seventy-three of the isolated compounds exhibited a phenolic or benzylic structure. Fifty-four compounds were reported for the first time as Riesling wine constituents. New compounds identified in this work included twelve benzoic and cinnamic acid derivatives. In addition to two isomeric (E)-caffeoyl ethyl tartrates, the glucose esters of (E)-cinnamic, (E)-p-coumaric, and (E)-ferulic acid, as well as the 4-O-glucosides of (E)- and (Z)-ferulic acid, have been identified for the first time in Riesling wine. The structures of two additional phenylpropanoids were elucidated as 3-hydroxy-1-(4-hydroxy-3-methoxyphenyl)-propan-1-one and 2,3-dihydroxy-1-(4-hydroxy-3-methoxyphenyl)-propan-1-one. Moreover, two ethyl esters, i.e., ethyl protocatechuate and ethyl gallate, as well as the glucose ester of vanillic acid, were newly detected in Riesling wine. Novel representatives in the flavonoid group were dihydrokaempferol, dihydroquercetin, and four dihydroflavonol glycoconjugates, i.e., the 3-O-glucosides of dihydrokaempferol and dihydroquercetin, as well as the 3-O-xyloside and the 3'-O-glucoside of dihydroquercetin. Additionally, six novel lignans, i.e., Lariciresinol 4-O-glucoside, three isolariciresinol derivatives, and two secoisolariciresinols, as well as three neolignans were isolated. Structural elucidation of the newly isolated wine constituents is reported together with the determination of their antioxidant activity.