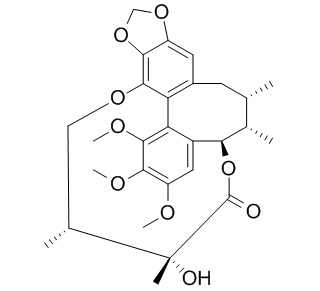
Gomisin E
Gomisin E is a natural product from Schizandra chinensis.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Biomed Pharmacother.2024, 179:117346.
Oxid Med Cell Longev2020, 12
Natural Product Communications2020, doi: 10.1177.
Food and Bioprocess Technology2017, 10(6):1074-1092
Int J Mol Sci.2021, 22(16):8641.
Key Engineering Materials2022, 931(47-53).
Biomed Pharmacother.2024, 175:116770.
Eur J Pharmacol.2024, 963:176280.
Phytomedicine.2023, 114:154813.
J Sci Food Agric.2017, 97(5):1656-1662
Related and Featured Products
J Sep Sci. 2012 Sep;35(17):2203-9.
Application of ultrahigh-performance liquid chromatography coupled with mass spectrometry for analysis of lignans and quality control of Fructus Schisandrae chinensis.[Pubmed:
22814992 ]
Lignans in the drug Fructus Schisandrae chinensis (FSC) exhibit potent biological activities such as antihepatotoxic, antiasthmatic, and antigastric ulcer.
METHODS AND RESULTS:
An ultrahigh-performance liquid chromatography coupled with quadrupole time-of-flight mass spectrometry method has been developed to evaluate the quality of FSC through simultaneous qualitative and quantitative analysis of 15 lignans, including schizandrin A, B, and C; schizandrol A and B; gomisin B, gomisin C, gomisin D, Gomisin E, gomisin G, gomisin H, gomisin J, and gomisin N; tigloylgomisin H; and angeloylgomisin H. The compounds were separated on a Zorbax Eclipse Plus C(18) (2.1 × 100 mm, 1.8 μm) column with a gradient elution of acetonitrile and 0.1% formic acid. Lignans were identified through their retention times, accurate mass data, and characteristic ions by comparison with a reference substance. All calibration curves showed perfect linear regression (r(2) > 0.99) within the test range. The limits of detection and quantitation fell in the ranges of 0.1-4 ng/mL for all the analytes with an injection of 10 μL.
CONCLUSIONS:
Good results were obtained with respect to repeatability (relative standard deviation <4.6%) and recovery (85.58-105.82%). Meanwhile, the entire sample analysis time was less than 10 min. This developed method provided a new basis for the overall assessment of the quality of FSC.