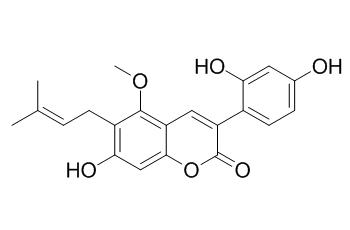
Glycycoumarin
Glycycoumarin is an estrogen agonist, it shows moderate inhibitory effects against CYP1A2 and CYP2B6. Glycycoumarin has anti-liver cancer, neuroprotective, antioxidant and anti-inflammatory activities. Glycycoumarin has anti-hepatitis C virus (HCV) activity, with the IC(50) value of 8.8, ug/mL. Glycycoumarin has an inhibitory effect on smooth muscle contraction induced by various types of stimulants through the inhibition of PDEs, especially isozyme 3, followed by the accumulation of intracellular cAMP; it also can inhibit hepatocyte lipoapoptosis through activation of autophagy and inhibition of ER stress/GSK-3-mediated mitochondrial pathway.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Int. J. Mol. Sci. 2022, 23(3),1696.
Pharmacol Res.2022, 182:106346.
Acta Chromatographica2021, 00960.
Molecules.2023, 28(19):6775.
Phytother Res.2022, 35844057.
Journal of Medicinal Food2023, Vol.26(10).
Sci Rep.2021, 11(1):10931.
Sci Rep. 2024, 14(1):70.
Environ Toxicol.2021, doi: 10.1002
BMC Complement Altern Med.2019, 19(1):11
Related and Featured Products
Oncotarget. 2016 Oct 4;7(40):65732-65743.
Glycycoumarin exerts anti-liver cancer activity by directly targeting T-LAK cell-originated protein kinase.[Pubmed:
27582549 ]
Glycycoumarin (GCM) is a major bioactive coumarin compound isolated from licorice and the anti-cancer activity of GCM has not been scientifically addressed.
METHODS AND RESULTS:
In the present study, we have tested the anti-liver cancer activity of GCM using both in vitro and in vivo models and found for the first time that GCM possesses a potent activity against liver cancer evidenced by cell growth inhibition and apoptosis induction in vitro and tumor reduction in vivo. Mechanistically, GCM was able to bind to and inactivate oncogenic kinase T-LAK cell-originated protein kinase (TOPK), which in turn led to activation of p53 pathway.
CONCLUSIONS:
Our findings supported GCM as a novel active compound that contributed to the anti-cancer activity of licorice and TOPK could be an effective target for hepatocellular carcinoma (HCC) treatment.
Food Chem. 2013 Nov 15;141(2):1063-71.
Antioxidant and anti-inflammatory activities of six flavonoids separated from licorice.[Pubmed:
23790887]
Licorice, the roots and rhizomes of several Glycyrrhiza species (Leguminosae), is an important natural sweetening agent and a widely used herbal medicine.
METHODS AND RESULTS:
In this work, six flavonoids, 5-(1,1-dimethylallyl)-3,4,4'-trihydroxy-2-methoxychalcone (1), licochalcone B (2), licochalcone A (3), echinatin (4), Glycycoumarin (5) and glyurallin B (6), were isolated from the extracts of licorice (Glycyrrhiza inflata and Glycyrrhiza uralensis). Their structures were elucidated using various spectroscopic methods. To our knowledge, compound 1 was isolated from natural plants for the first time. All the isolates were tested by antioxidant and anti-inflammatory assays. Compounds 2, 4 and 5 showed strong scavenging activity toward the ABTS(+) radical, and compounds 1, 2, 3, 5 and 6 exhibited potent inhibition of lipid peroxidation in rat liver microsomes compared with the reference controls. Compounds 1-4 dose-dependently inhibited LPS induced reactive oxygen species (ROS) production in RAW 264.7 cells. Furthermore, compounds 1-5 were demonstrated to inhibit the production of nitric oxide (NO), interleukin-6 (IL-6) and prostaglandin E2 (PGE2) in LPS-induced macrophage cells. Moreover, the contents of the six compounds, in different Glycyrrhiza species, were quantified by HPLC-MS.
Microbiol Immunol. 2014 Mar;58(3):180-7
Anti-hepatitis C virus compounds obtained from Glycyrrhiza uralensis and other Glycyrrhiza species.[Pubmed:
24397541 ]
Development of complementary and/or alternative drugs for treatment of hepatitis C virus (HCV) infection is still much needed from clinical and economic points of view. Antiviral substances obtained from medicinal plants are potentially good targets to study. Glycyrrhiza uralensis and G. glabra have been commonly used in both traditional and modern medicine.
METHODS AND RESULTS:
In this study, extracts of G. uralensis roots and their components were examined for anti-HCV activity using an HCV cell culture system. It was found that a methanol extract of G. uralensis roots and its chloroform fraction possess anti-HCV activity with 50%-inhibitory concentrations (IC(50)) of 20.0 and 8.0 μg/mL, respectively. Through bioactivity-guided purification and structural analysis, Glycycoumarin, glycyrin, glycyrol and liquiritigenin were isolated and identified as anti-HCV compounds, their IC(50) being 8.8, 7.2, 4.6 and 16.4 μg/mL, respectively. However, glycyrrhizin, the major constituent of G. uralensis, and its monoammonium salt, showed only marginal anti-HCV activity. It was also found that licochalcone A and glabridin, known to be exclusive constituents of G. inflata and G. glabra, respectively, did have anti-HCV activity, their IC(50) being 2.5 and 6.2 μg/mL, respectively. Another chalcone, isoliquiritigenin, also showed anti-HCV activity, with an IC(50) of 3.7 μg/mL. Time-of-addition analysis revealed that all Glycyrrhiza-derived anti-HCV compounds tested in this study act at the post-entry step.
CONCLUSIONS:
In conclusion, the present results suggest that Glycycoumarin, glycyrin, glycyrol and liquiritigenin isolated from G. uralensis, as well as isoliquiritigenin, licochalcone A and glabridin, would be good candidates for seed compounds to develop antivirals against HCV.
Sci Rep . 2016 Nov 30;6:38138.
Glycycoumarin inhibits hepatocyte lipoapoptosis through activation of autophagy and inhibition of ER stress/GSK-3-mediated mitochondrial pathway[Pubmed:
27901086]
Abstract
Herbal medicine as an alternative approach in the treatment of disease has drawn growing attention. Identification of the active ingredient is needed for effective utilization of the herbal medicine. Licorice is a popular herbal plant that is widely used to treat various diseases including liver diseases. Glycycoumarin (GCM) is a representative of courmarin compounds isolated from licorice. In the present study, the protective effect of GCM on hepatocyte lipoapoptosis has been evaluated using both cell culture model of palmitate-induced lipoapoptosis and animal model of non-alcoholic steatohepatitis (NASH). The results demonstrated for the first time that GCM was highly effective in suppressing hepatocyte lipoapoptosis in both in vitro and in vivo. Mechanistically, GCM was able to re-activate the impaired autophagy by lipid metabolic disorders. In line with the activation of autophagy, ER stress-mediated JNK and mitochondrial apoptotic pathway activation was inhibited by GCM both in vitro and in vivo. In addition, inactivation of GSK-3 might also contribute to the protective effect of GCM on hepatocyte lipoapoptosis. Our findings supported GCM as a novel active component of licorice against non-alcoholic fatty liver disease (NAFLD).
Eur J Pharm Sci. 2017 Nov 15;109:182-190.
Cytochrome P450 inhibition by three licorice species and fourteen licorice constituents.[Pubmed:
28774812]
METHODS AND RESULTS:
The potential of licorice dietary supplements to interact with drug metabolism was evaluated by testing extracts of three botanically identified licorice species (Glycyrrhiza glabra L., Glycyrrhiza uralensis Fish. ex DC. and Glycyrrhiza inflata Batalin) and 14 isolated licorice compounds for inhibition of 9 cytochrome P450 enzymes using a UHPLC-MS/MS cocktail assay. G. glabra showed moderate inhibitory effects against CYP2B6, CYP2C8, CYP2C9, and CYP2C19, and weak inhibition against CYP3A4 (testosterone). In contrast, G. uralensis strongly inhibited CYP2B6 and moderately inhibited CYP2C8, CYP2C9 and CYP2C19, and G. inflata strongly inhibited CYP2C enzymes and moderately inhibited CYP1A2, CYP2B6, CYP2D6, and CYP3A4 (midazolam). The licorice compounds isoliquiritigenin, licoricidin, licochalcone A, 18β-glycyrrhetinic acid, and Glycycoumarin inhibited one or more members of the CYP2C family of enzymes. Glycycoumarin and licochalcone A inhibited CYP1A2, but only Glycycoumarin inhibited CYP2B6. Isoliquiritigenin, glabridin and licoricidin competitively inhibited CYP3A4, while licochalcone A (specific to G. inflata roots) was a mechanism-based inhibitor. The three licorice species commonly used in botanical dietary supplements have varying potential for drug-botanical interactions as inhibitors of cytochrome P450 isoforms.
CONCLUSIONS:
Each species of licorice displays a unique profile of constituents with potential for drug interactions.
Steroids. 2016 Jan;105:42-9.
Licorice root components in dietary supplements are selective estrogen receptor modulators with a spectrum of estrogenic and anti-estrogenic activities.[Pubmed:
26631549 ]
Licorice root extracts are often consumed as botanical dietary supplements by menopausal women as a natural alternative to pharmaceutical hormone replacement therapy. In addition to their components liquiritigenin (Liq) and isoliquiritigenin (Iso-Liq), known to have estrogenic activity, licorice root extracts also contain a number of other flavonoids, isoflavonoids, and chalcones.
METHODS AND RESULTS:
We have investigated the estrogenic activity of 7 of these components, obtained from an extract of Glycyrrhiza glabra powder, namely Glabridin (L1), Calycosin (L2), Methoxychalcone (L3), Vestitol (L4), Glyasperin C (L5), Glycycoumarin (L6), and Glicoricone (L7), and compared them with Liq, Iso-Liq, and estradiol (E2). All components, including Liq and Iso-Liq, have low binding affinity for estrogen receptors (ERs). Their potency and efficacy in stimulating the expression of estrogen-regulated genes reveal that Liq and Iso-Liq and L2, L3, L4, and L6 are estrogen agonists. Interestingly, L3 and L4 have an efficacy nearly equivalent to E2 but with a potency ca. 10,000-fold less. The other components, L1, L5 and L7, acted as partial estrogen antagonists. All agonist activities were reversed by the antiestrogen, ICI 182,780, or by knockdown of ERα with siRNA, indicating that they are ER dependent. In HepG2 hepatoma cells stably expressing ERα, only Liq, Iso-Liq, and L3 stimulated estrogen-regulated gene expression, and in all cases gene stimulation did not occur in HepG2 cells lacking ERα.
CONCLUSIONS:
Collectively, these findings classify the components of licorice root extracts as low potency, mixed ER agonists and antagonists, having a character akin to that of selective estrogen receptor modulators or SERMs.
J Ethnopharmacol. 2006 May 24;105(3):409-14.
Glycycoumarin from Glycyrrhizae Radix acts as a potent antispasmodic through inhibition of phosphodiesterase 3.[Pubmed:
16387459 ]
Glycyrrhizae Radix is used to treat abdominal pain as a component of Shakuyaku-kanzo-to, a traditional Chinese medicine formulation. We aim at clarifying the antispasmodic principles of Glycyrrhizae Radix, and consequently isolated Glycycoumarin as a potent relaxant on the carbamylcholine (CCh)-induced contraction of mouse jejunum.
METHODS AND RESULTS:
In this paper we investigated the effects and the action mechanism of Glycycoumarin on the contraction of mouse jejunum. Glycycoumarin inhibited the contraction induced by various types of stimulants, such as CCh, KCl, BaCl(2), and A23187 (calcium ionophore III) with IC(50) values of 2.93+/-0.94 micromol/l (1.08+/-0.35 microg/ml), 2.59+/-0.58 micromol/l (0.95+/-0.29 microg/ml), 4.09+/-1.82 micromol/l (1.51+/-0.67 microg/ml) and 7.39+/-5.19 micromol/l (2.72+/-1.91 microg/ml), respectively, with a potency similar to that of papaverine (a representative antispasmodic for smooth muscle). Furthermore, pretreatment with Glycycoumarin enhanced the relaxation induced by forskolin on CCh-evoked contraction, similar to that by pretreatment with IBMX, a non-specific inhibitor of phosphodiesterases (PDEs). Pretreatment with Glycycoumarin also enhanced the relaxation effect of rolipram, a specific inhibitor of PDE isozyme 4, as pretreatment with milrinone, a specific inhibitor of isozyme 3, did. Moreover, the effect of Glycycoumarin was associated with dose-dependent accumulation of cAMP, but not cGMP, in mouse jejunum.
CONCLUSIONS:
These results indicate that Glycycoumarin has an inhibitory effect on smooth muscle contraction induced by various types of stimulants through the inhibition of PDEs, especially isozyme 3, followed by the accumulation of intracellular cAMP.
Sci Rep. 2016 Nov 30;6:38138.
Glycycoumarin inhibits hepatocyte lipoapoptosis through activation of autophagy and inhibition of ER stress/GSK-3-mediated mitochondrial pathway.[Pubmed:
27901086 ]
Herbal medicine as an alternative approach in the treatment of disease has drawn growing attention. Identification of the active ingredient is needed for effective utilization of the herbal medicine. Licorice is a popular herbal plant that is widely used to treat various diseases including liver diseases. Glycycoumarin (GCM) is a representative of courmarin compounds isolated from licorice.
METHODS AND RESULTS:
In the present study, the protective effect of GCM on hepatocyte lipoapoptosis has been evaluated using both cell culture model of palmitate-induced lipoapoptosis and animal model of non-alcoholic steatohepatitis (NASH). The results demonstrated for the first time that GCM was highly effective in suppressing hepatocyte lipoapoptosis in both in vitro and in vivo. Mechanistically, GCM was able to re-activate the impaired autophagy by lipid metabolic disorders. In line with the activation of autophagy, ER stress-mediated JNK and mitochondrial apoptotic pathway activation was inhibited by GCM both in vitro and in vivo. In addition, inactivation of GSK-3 might also contribute to the protective effect of GCM on hepatocyte lipoapoptosis.
CONCLUSIONS:
Our findings supported GCM as a novel active component of licorice against non-alcoholic fatty liver disease (NAFLD).
J Ethnopharmacol. 2015 Jan 15;159:122-8.
Protective effects of glycycoumarin and procyanidin B1, active components of traditional Japanese medicine yokukansan, on amyloid β oligomer-induced neuronal death.[Pubmed:
25446602 ]
Yokukansan, a traditional Japanese (Kampo) medicine, is composed of seven medicinal herbs, and has been traditionally used to treat neurosis, insomnia, and night crying and irritability in children. Yokukansan and its constituent herbs, Glycyrrhiza and Uncaria Hook, have recently been shown to have protective effects against amyloid β (Aβ) oligomer-induced apoptosis by suppressing the activation of caspase-3 in primary cultured neurons. The aim of the present study was to identify the effective components of Glycyrrhiza and Uncaria Hook against Aβ oligomer-induced neurotoxicity. We also attempted to clarify the mechanisms by which yokukansan and these herbs, as well as their components, suppressed the activation of caspase-3 in Aβ oligomer-treated neurons.
METHODS AND RESULTS:
Rat primary cultured cortical neurons were treated with Aβ oligomer (3 μM). The protective effects of 16 components derived from Glycyrrhiza or Uncaria Hook against Aβ oligomer-induced neurotoxicity were determined using the MTT reduction assay 48 h after the treatment. The suppressive effects of the test substances, i.e., yokukansan, Glycyrrhiza, Uncaria Hook, and screened components, on the Aβ oligomer-induced activation of caspase-3(/7) were evaluated using the caspase-Glo assay 48 h after the Aβ oligomer treatment. The suppressive effects of the test substances on the activation of caspase-8 and -9, both of which are located upstream of caspase-3, were also examined 24h after the Aβ oligomer treatment.
Two of the 16 components tested, Glycycoumarin derived from Glycyrrhiza and procyanidin B1 derived from Uncaria Hook, significantly inhibited Aβ oligomer-induced neuronal death in a dose-dependent manner. Glycyrrhiza, Uncaria Hook, and yokukansan significantly suppressed the Aβ oligomer-induced activation of caspase-3 as well as caspase-8 and -9. Glycycoumarin also suppressed the activation of caspase-3, but not caspase-8 and -9. Procyanidin B1 suppressed the activation of caspase-3, -8, and -9.
CONCLUSIONS:
Our results demonstrated that Glycycoumarin and procyanidin B1 had ameliorative effects on Aβ oligomer-induced neurotoxicity. The neuroprotective effects of Glycycoumarin are thought to be due to the attenuated activation of caspase-3, but not caspase-8 or -9. Procyanidin B1, as well as yokukansan, Glycyrrhiza, and Uncaria Hook, may attenuate the activation of caspase-3 by inhibiting that of caspase-8 and -9.