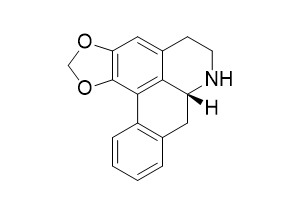
(-)-Anonaine
(-)-Anonaine has some anticancer activity, it induces apoptosis through Bax- and caspase-dependent pathways in human cervical cancer (HeLa) cells, it induces DNA damage and inhibits growth and migration of human lung carcinoma h1299 cells. (-)-Anonaine may be considered a potent compound for chemotherapy against cervical cancer or a health food supplement for cancer chemoprevention.
(-)-Anonaine has vasorelaxant effect.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Molecules.2024, 29(23):5632.
Nutrients.2021, 13(1):254.
J Sci Food Agric.2024, 104(7):4425-4437.
Microchemical Journal2018, 137:168-173
Int J Mol Sci.2023, 24(6):5769.
Processes2021, 9(1), 153.
Phytomedicine2022, 104:154318
Asian J Beauty Cosmetol2020, 18(3): 265-272.
Molecules.2016, 21(6)
Kor. J. Herbol.2019, 34(2):59-66
Related and Featured Products
Planta Med. 2004 Jul;70(7):603-9.
Vascular activity of (-)-anonaine, (-)-roemerine and (-)-pukateine, three natural 6a(R)-1,2-methylenedioxyaporphines with different affinities for alpha1-adrenoceptor subtypes.[Pubmed:
15254852 ]
We have studied the mechanism of action of three 6a( R)-1,2-methylenedioxyaporphines as vasorelaxant compounds.
METHODS AND RESULTS:
The alkaloids assayed showed different affinities for the three human cloned alpha (1)-adrenoceptor (AR) subtypes stably expressed in rat-1 fibroblasts, showing lower affinity for alpha(1B)-AR with regard to the alpha(1A)- or alpha(1D)-subtypes. These three natural compounds are more potent inhibitors of [ (3)H]-prazosin binding than of [ (3)H]-diltiazem binding to rat cerebral cortical membranes. As all these alkaloids inhibited noradrenaline (NA)-induced [ (3)H]-inositol phosphate formation in cerebral cortex and rat tail artery, they may be safely viewed as alpha (1)-AR antagonists, as is demonstrated by the vasorelaxant responses observed in isolated rat tail artery and/or aorta precontracted with NA. The alkaloids also inhibited the contractile response evoked by KCl (80 mM) but with a lower potency than that shown against NA-induced contraction. We have also examined their ability to inhibit the different forms of cyclic nucleotide phosphodiesterases (PDE) isolated from bovine aortic smooth muscle and endothelial cells, with negative results.
CONCLUSIONS:
We conclude that N-methylation favours the interaction of (R)-aporphines with all alpha (1)-AR subtypes, and that the topography of the binding site recognizing the basic or protonated nitrogen atom is similar in all three alpha (1)-AR subtypes. The presence of a hydroxy group at C-11 has different effects on the affinity for each alpha (1)-AR subtype but decreases the affinity for Ca (2+) channels. These results confirm and extend the view that subtle changes in the hydroxylation patterns on the aromatic ring of the aporphine structure affect the interactions of these compounds with the three alpha (1)-AR subtypes in different ways, suggesting that the binding site recognizing the aporphine skeleton is different in each of the three subtypes..
Food Chem Toxicol. 2008 Aug;46(8):2694-702.
(-)-Anonaine induces apoptosis through Bax- and caspase-dependent pathways in human cervical cancer (HeLa) cells.[Pubmed:
18524447 ]
(-)-Anonaine has been shown to have some anticancer activities, but the mechanisms of (-)-Anonaine inducing cell death of human cancer cells is not fully understood. We investigated the mechanisms of apoptosis induced by (-)-Anonaine in human HeLa cancer cells.
METHODS AND RESULTS:
Treatment with (-)-Anonaine induces dose-dependent DNA damage that is correlated with increased intracellular nitric oxide, reactive oxygen species, glutathione depletion, disruptive mitochondrial transmembrane potential, activation of caspase 3, 7, 8, and 9, and poly ADP ribose polymerase cleavage. Our data indicate that (-)-Anonaine up-regulated the expression of Bax and p53 proteins in HeLa cancer cells. The apoptosis and expression of Bax induced by (-)-Anonaine could be inhibited when the HeLa cells were pretreated with Boc-Asp(OMe)-fmk, which is a broad caspases inhibitor. There was no obvious DNA damage in the (-)-Anonaine-treated Madin-Darby canine kidney and Vero cell lines. Both Madin-Darby canine kidney and Vero cell lines are kidney epithelial cellular morphology.
CONCLUSIONS:
These results suggest that (-)-Anonaine might be considered a potent compound for chemotherapy against cervical cancer or a health food supplement for cancer chemoprevention.
J Agric Food Chem. 2011 Mar 23;59(6):2284-90.
(-)-Anonaine induces DNA damage and inhibits growth and migration of human lung carcinoma h1299 cells.[Pubmed:
21361287]
The anticancer effects of (-)-Anonaine were investigated in this current study.
METHODS AND RESULTS:
(-)-Anonaine at concentration ranges of 50-200 μM exhibited significant inhibition to cell growth and migration activities on human lung cancer H1299 cells at 24 h, albeit cell cycle analyses showed that (-)-Anonaine at the above concentration ranges did not cause any significant changes in cell-cycle distributions. Significant nuclear damages of H1299 cells were observed with 10-200 μM (-)-Anonaine treatment in a comet assay, whereas higher concentrations (6 and 30 mM) of (-)-Anonaine concentrations were required to cause DNA damages in an in vitro plasmid cleavage assay.
CONCLUSIONS:
In summary, our results demonstrated that (-)-Anonaine exhibited dose-dependent antiproliferatory, antimigratory, and DNA-damaging effects on H1299 cells. We inferred that (-)-Anonaine can cause cell-cycle arrest and DNA damage to hamper the physiological behavior of cancer cells at 72 h, and therefore, it can be useful as one of the potential herbal supplements for chemoprevention of human lung cancer.