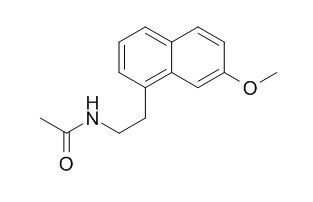
Agomelatine
Agomelatine is a competitive antagonist of human and porcine serotonin (5-HT2C) receptors (pKi = 6.2 and 6.4, respectively) as well as human 5-HT2B receptors (pKi = 6.6). Agomelatine is a melatonin (MT) analogue with agonistic properties and has been proven to be effective for various types of depressive symptoms, agomelatine also has anticonvulsant activity. Agomelatine treatment could represent a novel useful approach to the clinical care of subjects with Chronic Fatigue Syndrome (CFS). Agomelatine administration protects liver cells from paracetamol-induced hepatotoxicity via antioxidant activity and reduced proinflammatory cytokines, such as TNF-α and IL-6.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Biomed Pharmacother.2022, 146:112497.
Applied Biological Chem. 2020, 26(63).
Phytother Res.2018, 32(5):923-932
Pathogens.2018, 7(3):E62
Int J Mol Sci.2024, 25(17):9673.
Int J Mol Med.2016, 37(2):501-8
Biol. Life Sci. Forum2024, 35(1), 6.
Heliyon2020, 6(6):e04337.
Plant Archives2020, 2(1),2929-2934
Postharvest Biol Tec2019, 149:18-26
Related and Featured Products
Hum Exp Toxicol. 2013 Aug;32(8):846-57.
Agomelatine: an antidepressant with new potent hepatoprotective effects on paracetamol-induced liver damage in rats.[Pubmed:
23584358]
Paracetamol was shown to induce hepatotoxicity or more severe fatal acute hepatic damage. Agomelatine, commonly known as melatonin receptor agonist, is a new antidepressant, which resynchronizes circadian rhythms with subjective and objective improvements in sleep quality and architecture, as melatonin does.
METHODS AND RESULTS:
In the present study, it was aimed to evaluate the hepatoprotective activity of Agomelatine on paracetamol-induced hepatotoxicity and to understand the relationship between the hepatoprotective mechanism of Agomelatine and antioxidant system and proinflammatory cytokines. A total of 42 rats were divided into 7 groups as each composed of 6 rats: (1) intact, (2) 40 mg/kg Agomelatine, (3) 140 mg/kg N-acetylcysteine (NAC), (4) 2 g/kg paracetamol, (5) 2 g/kg paracetamol + 140 mg/kg NAC, (6) 2 g/kg paracetamol + 20 mg/kg Agomelatine, and (7) 2 g/kg paracetamol + 40 mg/kg Agomelatine groups. Paracetamol-induced hepatotoxicity was applied and liver and blood samples were analyzed histopathologically and biochemically. There were statistically significant increases in the activities of aspartate aminotransferase, alanine aminotransferase, levels of tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6) and 8-iso-prostane, and decreases in the activity of superoxide dismutase and level of glutathione in the group treated with paracetamol. Administration of Agomelatine and NAC separately reversed these changes significantly.
CONCLUSIONS:
In conclusion, Agomelatine administration protects liver cells from paracetamol-induced hepatotoxicity via antioxidant activity and reduced proinflammatory cytokines, such as TNF-α and IL-6.
Eur Neuropsychopharmacol. 2014 Jun;24(6):939-44.
Agomelatine but not melatonin improves fatigue perception: a longitudinal proof-of-concept study.[Pubmed:
24636462]
Chronic Fatigue Syndrome (CFS) represents a disabling condition characterized by persistent mental and physical fatigue, bodily discomfort and cognitive difficulties. To date the neural bases of CFS are poorly understood; however, mono-aminergic abnormalities, sleep-wake cycle changes and prefrontal dysfunctions are all thought to play a role in the development and maintenance of this condition.
METHODS AND RESULTS:
Here we explored in a group of 62 CFS subjects the impact on fatigue levels of Agomelatine, an antidepressant with agonist activity at melatonin receptors (MT1 and MT2) and antagonist activity at serotoninergic 2C receptors (5HT2C). To tease out the relative effects of MT-agonism and 5HT2C antagonism on fatigue, we compared Agomelatine 50mg u.i.d. with sustained release melatonin 10mg u.i.d. in the first 12-week-long phase of the study, and then switched all melatonin-treated subjects to Agomelatine in the second 12-week-long phase of the study. Agomelatine treatment, but not melatonin, was associated with a significant reduction of perceived fatigue and an increase in perceived quality of life. Moreover the switch from melatonin to Agomelatine was associated with a reduction of fatigue levels. Agomelatine was well tolerated by all enrolled subjects.
CONCLUSIONS:
Our data, albeit preliminary, suggest that Agomelatine treatment could represent a novel useful approach to the clinical care of subjects with CFS.
Expert Opin Drug Metab Toxicol. 2014 Jun;10(6):885-92.
Pharmacokinetic evaluation of agomelatine for the treatment of generalised anxiety disorder.[Pubmed:
24717138]
Preliminary data indicate Agomelatine as a promising molecule for both acute and long-term treatment of generalised anxiety disorder (GAD).
METHODS AND RESULTS:
The present review illustrates the pharmacokinetic properties of Agomelatine and their implications for the management of GAD patients. A search of the main database sources (Medline, Isi Web of Knowledge and Medscape) was performed in order to obtain a complete and balanced evaluation of Agomelatine pharmacokinetics for the treatment of GAD. The word 'Agomelatine' was associated with 'pharmacokinetics', 'GAD', 'anxiety' and 'tolerability'. No restriction criteria were established in relation to methodology or year of publication. Only English-language articles were included.
CONCLUSIONS:
Short half-life and 1-day administration make Agomelatine an interesting molecule for GAD treatment. However, potential interactions with a number of compounds necessitate caution when prescribing and using Agomelatine in patients with psychiatric (e.g., alcohol abuse) or medical comorbidities. Further data are necessary to define a precise risk/benefit ratio in special populations such as elderly patients suffering from GAD.
J Clin Pharm Ther. 2014 Apr;39(2):204-9.
Effect of CYP1A2 polymorphism on the pharmacokinetics of agomelatine in Chinese healthy male volunteers.[Pubmed:
24372004]
Agomelatine is a melatonin (MT) analogue with agonistic properties and has been proven to be effective for various types of depressive symptoms. Following oral administration, Agomelatine is primarily metabolized by the hepatic cytochrome P450 isoenzyme CYP1A2. The purpose of this study was to assess the influence of CYP1A2 single nucleotide polymorphisms (SNPs, rs762551, rs2069514, rs2472304, rs2470890) on Agomelatine pharmacokinetics in the Chinese population.
METHODS AND RESULTS:
Seventy-two healthy Chinese male volunteers enrolled in the study received an oral dose of 25 mg of Agomelatine after providing written informed consent. CYP1A2 SNPs were genotyped by polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP). Agomelatine plasma concentrations were determined by high performance liquid chromatography-tandem mass spectrometry, and the pharmacokinetics analyses were evaluated by nonparametric methods. After a single oral dose of 25 mg Agomelatine, no significant differences existed in Agomelatine pharmacokinetics between the rs2069514 GG homozygotes (n = 35) and the rs2069514 AG allele (n = 35) in all subjects. The mean Agomelatine AUC0-7 , AUC0-∞ and Cmax for the rs762551 CC homozygotes (n = 9), rs2470890 CC homozygotes (n = 54) and rs2472304 GG homozygotes (n = 51) were much higher than the rs762551 AA allele (n = 31), rs2470890 CT allele (n = 17) and rs2472304 AG allele (n = 20) respectively (P < 0.05).
CONCLUSIONS:
The rs762551 A, rs2470890 T and rs2472304 A genotype presented a significantly lower level of Agomelatine exposure (AUC, Cmax ) compared with the rs762551 C, rs2470890 C and rs2472304 G genotype in Chinese healthy subjects. It suggested that the rs762551, rs2470890 and rs2472304 genetic polymorphism might be associated with the marked interindividual variability of Agomelatine, and the pharmacokinetic profile of Agomelatine may be different in different races.
Epilepsy Behav. 2012 Jul;24(3):324-8.
Anticonvulsant effects of agomelatine in mice.[Pubmed:
22658946]
Agomelatine is a potent MT1 and MT2 melatonin receptor agonist and a 5-HT2C serotonin receptor antagonist.
METHODS AND RESULTS:
We analyzed whether Agomelatine has anticonvulsant properties. The anticonvulsant activity of Agomelatine (25, 50 or 75 mg/kg, i.p.) was evaluated in mouse models of pentylenetetrazole (PTZ-85 mg/kg, i.p.), pilocarpine (400mg/kg, i.p.), picrotoxin (7 mg/kg, i.p.), strychnine (75 mg/kg, i.p.) or electroshock-induced convulsions. In the PTZ-induced seizure model, Agomelatine (at 25 or 50mg/kg) showed a significant increase in latency to convulsion, and Agomelatine (at 50 or 75 mg/kg) also increased significantly time until death. In the pilocarpine-induced seizure model, only Agomelatine in high doses (75 mg/kg) showed a significant increase in latency to convulsions and in time until death. In the strychnine-, electroshock- and picrotoxin-induced seizure models, Agomelatine caused no significant alterations in latency to convulsions and in time until death when compared to controls.
CONCLUSIONS:
Our results suggest that Agomelatine has anticonvulsant activity shown in PTZ- or pilocarpine-induced seizure models.
Ganoderic acid L
Catalog No: CFN95022
CAS No: 102607-24-9
Price: $413/5mg
Secologanic acid
Catalog No: CFN95028
CAS No: 60077-46-5
Price: $318/20mg
Gancaonin N
Catalog No: CFN95066
CAS No: 129145-52-4
Price: $463/5mg
7,2',4'-Trihydroxy-5-methoxy-3-arylcoumarin
Catalog No: CFN95070
CAS No: 1092952-62-9
Price: Inquiry(manager@chemfaces.com)
5,7,2',4'-Tetrahydroxy-8,3'-di(gamma,gamma-dimethylallyl)-isoflavanone
Catalog No: CFN95084
CAS No: 141846-47-1
Price: $413/5mg
Polygalasaponin XXXV
Catalog No: CFN95176
CAS No: 184479-28-5
Price: $463/5mg
2-Methoxy-1,4-naphthoquinone
Catalog No: CFN95269
CAS No: 2348-82-5
Price: $70/20mg
Euphorbia factor L24
Catalog No: CFN95334
CAS No: 1613700-13-2
Price: $318/5mg
Hydroxytyrosol 1-O-glucoside
Catalog No: CFN95367
CAS No: 76873-99-9
Price: $318/10mg
2-(2,4-Dihydroxyphenyl)-5,6-methylenedioxybenzofuran (ABF)
Catalog No: CFN95511
CAS No: 67121-26-0
Price: $318/5mg