Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
J Nat Prod. 2004 Dec;67(12):1980-4.
New furofuran and butyrolactone lignans with antioxidant activity from the stem bark of Styrax japonica.[Pubmed:
15620237]
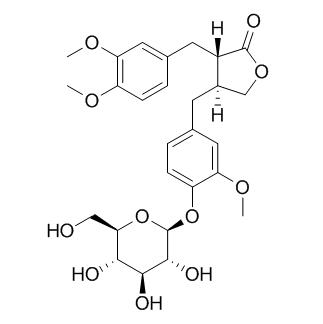
A new furofuran lignan, styraxlignolide B (1), and four new dibenzyl-gamma-butyrolactone lignans, styraxlignolides C, styraxlignolide D, styraxlignolide E, and Styraxlignolide F (2-5), were isolated from the EtOAc-soluble fraction of stem bark of Styrax japonica.
METHODS AND RESULTS:
Known compounds, taraxerol (6), syringin (7), and (-)-pinoresinol glucoside (8), were also obtained. The structures of styraxligonolides B-F were determined as 2alpha-(4'-hydroxy-3'-methoxyphenyl)-6alpha-(3' ',4' '-methylenedioxyphenyl)-8-oxo-3,7-dioxabicyclo[3.3.0]octane 4'-O-(beta-D-glucopyranoside) (1), (2S,3S)-2alpha-(3' '-hydroxy-4' '-methoxybenzyl)-3beta-(4'-hydroxy-3'-methoxybenzyl)-gamma-butyrolactone 4'-O-(beta-D-glucopyranoside) (2), (2S,3S)-2alpha-(4' '-hydroxy-3' '-methoxybenzyl)-3beta-(4'-hydroxy-3'-methoxybenzyl)-gamma-butyrolactone 4'-O-(beta-D-glucopyranoside) (3), (2S,3S)-2alpha-(4' '-hydroxy-3' '-methoxybenzyl)-3beta-(4'-hydroxy-3'-methoxybenzyl)-gamma-butyrolactone 4' '-O-(beta-D-glucopyranoside) (4), and (2S,3S)-2alpha-(3' ',4' '-dimethoxybenzyl)-3beta-(4'-hydroxy-3'-methoxybenzyl)-gamma-butyrolactone 4'-O-(beta-D-glucopyranoside) (5) by spectroscopic means including 2D NMR.
CONCLUSIONS:
Compounds 1-8 were tested in vitro for antioxidant activity against 1,1-diphenyl-2-picrylhydrazyl (DPPH) radicals. Styraxlignolide C (2), styraxlignolide D (3), styraxlignolide E (4), and (-)-pinoresinol glucoside (8) exhibited weak radical-scavenging activity in the DPPH assay, with IC50 values of 380, 278, 194, and 260 microM, respectively.