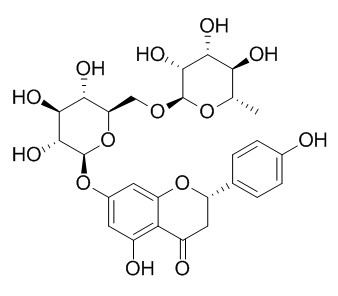
Narirutin
Citrus narirutin fraction (CNF), contained 75% of narirutin, co-administration of CNF with alcohol can alleviate alcohol induced liver damage through preventing lipid formation, protecting antioxidant system and suppressing productions of pro-inflammatory cytokines. Narirutin has anti-inflammatory effect in a murine model of allergic eosinophilic airway inflammation, the mechanism is likely to be associated with a reduction in the OVA-induced increases of IL-4 and IgE.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
International. J. of Food Properties 2017, 20:S131-S140
Molecules.2020, 25(23):5609.
Biomed Pharmacother.2024, 176:116765.
Biomed Pharmacother.2020, 128:110318.
Molecules.2021, 26(4):816.
J Chem Inf Model.2021, 61(11):5708-5718.
Food Science&Tech. Res.2022, 28(2):123-132.
Eur J Pharmacol.2024, 978:176749.
Journal of Functional Foods2023, 104:105542
Oncol Rep.2021, 46(2):166.
Related and Featured Products
Food Chem Toxicol. 2012 Oct;50(10):3498-504.
Narirutin fraction from citrus peels attenuates LPS-stimulated inflammatory response through inhibition of NF-κB and MAPKs activation.[Pubmed:
22813871]
METHODS AND RESULTS:
In this study, we examined the regulatory activity of Narirutin fraction from citrus peels on the production of inflammatory mediators managing acute or chronic inflammatory diseases in macrophages. Narirutin fraction inhibited the release, by lipopolysaccharide (LPS)-stimulated macrophages, of nitric oxide (NO) and prostaglandin E2 (PGE2) through suppressing the expression of inducible NO synthase (iNOS) and cyclooxygenase-2 (COX-2), respectively. The release, by LPS stimulated macrophages, of interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF-α) was also reduced by Narirutin fraction in a dose-dependent manner. Furthermore, Narirutin fraction inhibited the LPS-mediated activation of nuclear factor-κB (NF-κB) and mitogen-activated protein kinases (MAPKs), which are signaling molecules involved in production of pro-inflammatory factors.
CONCLUSIONS:
As a result of these properties, Narirutin fraction has the potential to be used as a functional dietary supplement and effective anti-inflammatory agent.
Curr Comput Aided Drug Des . 2020;16(5):523-529.
Structure-based Discovery of Narirutin as a Shikimate kinase Inhibitor with Anti-tubercular Potency[Pubmed:
31654517]
Abstract
Background: Shikimate pathway is essential for tubercular bacillus but it is absent in mammals. Therefore, Shikimate kinase and other enzymes in the pathway are potential targets for the development of novel anti-tuberculosis drugs.
Objective: In the present study, Shikimate kinase is selected as the target for in silico screening of phytochemicals with an aim to discover a novel herbal drug against Mycobacterium tuberculosis (Mtb).
Methods: A structure-based drug discovery approach is undertaken for the execution of the objective. Virtual screening of phytochemical database NPACT against the target, Shikimate kinase (PDB ID 3BAF), is carried out followed by toxicity and drug-likeness filtration. Finally, a lead, Narirutin was selected for in vitro anti-tubercular study.
Results: Narirutin, present in citrus fruits, emerges as the lead. It is considered to be non-toxic with predicted high LD50 value, 12000 mg/kg body weight. The phytochemical is tested for its antitubercular activity in vitro. It has MIC99 62.5 μg/mL against the MtbH37Rv strain.
Conclusion: This is the first-ever report to show anti-tuberculosis potency of Narirutin.
Keywords: Molecular docking; Mycobacterium tuberculosis; drug-likeness; Narirutin; shikimate kinase; toxicity.
Food Chem Toxicol. 2013 May;55:637-44.
Narirutin fraction from citrus peels attenuates alcoholic liver disease in mice.[Pubmed:
23416143]
METHODS AND RESULTS:
This study aimed to demonstrate protective activities of the Narirutin fraction from peels of Citrus unshiu against ethanol-induced hepatic damage through an animal study. Citrus Narirutin fraction (CNF), contained 75% of Narirutin, was obtained by an ultra-sonicated extraction and further purification. ICR mice were divided into four groups; normaldiet control, ethanol control (6.5g ethanol/kg), low-CNF (ethanol+150mg CNF/kg) and high-CNF (ethanol+300mg CNF/kg) groups. Consumption of alcohol for 8weeks induced severe liver damage with increases in prognostic indicators such as aspartate transaminase, alanine transaminase in serum whereas co-administration of CNF suppressed their increases. Excessive accumulations in liver TG and TC in ethanol control group were also suppressed by co-administration of CNF. Co-administration of CNF maintained SOD activity, GSH and malondialdehyde levels close to those of the normal diet group. Chronic consumption of alcohol also stimulated abrupt increases in pro-inflammatory cytokines such as nuclear factor (NF)-κB, tumor necrosis factor (TNF)-α and interleukin (IL)-1β in liver otherwise co-administration of CNF effectively suppressed production of these cytokines dose-dependently.
CONCLUSIONS:
These results indicate that co-administration of CNF with alcohol can alleviate alcohol induced liver damage through preventing lipid formation, protecting antioxidant system and suppressing productions of pro-inflammatory cytokines.
Clin Exp Pharmacol Physiol. 2007 Aug;34(8):766-70.
Narirutin inhibits airway inflammation in an allergic mouse model.[Pubmed:
17600554]
1. Flavonoids are naturally occurring compounds that possess anti-allergic, anti-inflammatory, antiproliferative and anti-oxidant properties.
METHODS AND RESULTS:
In the present study, we investigated whether the flavonoid Narirutin could reduce airway inflammation in ovalbumin (OVA)-sensitized/challenged NC/Nga mice, a model of allergic eosinophilic airway inflammation. 2. Mice were initially immunized intraperitoneally with OVA on Days 0 and 7 and then challenged with inhaled OVA on Days 14, 15 and 16. In addition, some mice received Narirutin orally at doses of 0.1, 1 or 10 mg/kg bodyweight daily on Days 7-16. 3. At 10 mg/kg, but not 0.1 or 1 mg/kg, Narirutin significantly diminished OVA-induced airway inflammation caused by infiltration of lung tissue with inflammatory and mucus-producing cells, as well as reduced eosinophil counts in the peripheral blood and bronchoalveolar lavage fluid (BALF), interleukin (IL)-4 levels in BALF and IgE levels in serum. 4. The mechanism of the anti-inflammatory effect of Narirutin are likely to be associated with a reduction in the OVA-induced increases of IL-4 and IgE in a murine model of allergic eosinophilic airway inflammation.
CONCLUSIONS:
These findings suggest that Narirutin may be an effective new tool in the treatment of bronchial asthma.
J AOAC Int. 2009 May-Jun;92(3):789-96.
Liquid chromatographic determination of narirutin and hesperidin in Zhi Ke (Citrus aurantium L.) in the form of the raw herb and of the dried aqueous extract.[Pubmed:
19610369]
A validated analytical method is reported for the analysis of Narirutin and hesperidin in Zhi Ke (Citrus aurantium L.) in the form of the dried raw herb and of the commercially prepared dried aqueous extract.
METHODS AND RESULTS:
The samples were extracted by sonication in methanol and the extract was analyzed by liquid chromatography-photodiode array (PDA) detection with identity confirmation by negative electrospray ionization-tandem mass spectrometry (MS). A C18 column was used with a methanol-water gradient mobile phase. Narirutin and hesperidin were quantified at 284 nm using the PDA detector. Using the MS detector, the Narirutin precursor ion with m/z 579 produced daughter ions with m/z 271 and 151. For hesperidin, the precursor ion with m/z 609 produced the m/z 301, 285, and 164 ions. The amounts of Narirutin and hesperidin found in the certified raw herb were 14.2 and 147.9 mg/g, respectively, and in the dried aqueous extract the amounts were 9.2 and 8.6 mg/g, respectively. For the raw herb, the average recovery across the three spike levels (50, 100, and 150%) for Narirutin and hesperidin were 110.7 and 94.5%, respectively.
CONCLUSIONS:
For the dried aqueous extract, the average recovery across the three spike levels for Narirutin and hesperidin were 85.8 and 98.9%, respectively.