Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
J Nat Prod . 2000 Jun;63(6):812-816.
New taxonomically significant sesquiterpenoids from Leontodon autumnalis[Pubmed:
10869207]
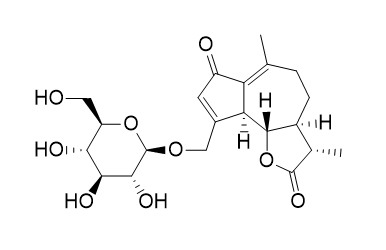
The methanolic extract of subaerial parts of Leontodon autumnalis afforded four new and two known sesquiterpenoids of the guaiane type. The known compounds were identified by means of (1)H and (13)C NMR spectroscopy as crepidiaside A (1) and B (2). The structures of the new compounds were determined by extensive 1D and 2D NMR experiments as 15-glucopyranosyloxy-2-oxo-guaia-3,11(13)-dien-1alp ha,5alpha, 6beta,7alpha,10alphaH-12,6-olide (3); 15-glucopyranosyloxy-2-oxo-guai-3-en-1alpha,5alpha+ ++,6beta,7alpha, 10alpha,11betaH-12,6-olide (4); 15-hydroxy-2-oxo-guai-3-en-1alpha, 5alpha,6beta,7alpha,10alpha,11betaH-12,6-+ ++olide (5); and 15-glucopyranosyloxy-2-oxo-guaia-3,11(13)-dien-1bet a,5alpha,6beta, 7alpha,10alphaH-12,6-olide (6), respectively. HPLC-DAD and HPLC-MS analyses of crude extracts of subaerial parts of 25 different taxa of the genus Leontodon revealed that compounds 1 and 2 occur in all investigated members of the section Oporinia (L. autumnalis, L. croceus, L. helveticus, L. montaniformis, L. montanus, L. pyrenaicus, and L. rilaensis) and in L. duboisii from the section Kalbfussia. Compounds 1-6 are detectable neither in other investigated taxa of Kalbfussia (L. cichoraceus, L. muelleri, and L. palisae) nor in any members of the subgenus Leontodon. Compounds 3-5 occur in high amounts only in L. croceus and L. pyrenaicus and in samples of L. autumnalis from northwestern Europe. In other members of the section Oporinia, in L. duboisii as well as in samples of L. autumnalis from the Pyrenees, the Alps, the Carpathians, and southern Central Europe, these substances occur only in trace amounts; in L. montanus and its closest relatives, compounds 3-5 are not detectable at all. Compound 6 is only detectable in samples of L. autumnalis, L. helveticus, L. pyrenaicus, L. rilaensis, and L. duboisii.