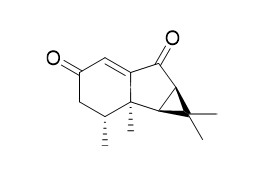
Nardoaristolone B
Nardoaristolone B, a nor-sesquiterpenoid with an unusual fused ring system and having protective effects on the injury of neonatal rat cardiomyocytes. The novel mosquito-repellentsynthetic hydrindanesbased on noreremophilanes and nardoaristolone B which show increasedactivity against adult females of Aedes aegypti.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
JPC-Journal of Planar Chromatography 2017, 30(2)
Pharmaceutics.2022, 14(3):564.
Int J Mol Sci.2024, 25(20):11227.
Separations2023, 10(7), 411.
Plant Cell Physiol.2023, 64(7):716-728.
Mol Med Rep.2024, 29(2):26.
Sci Rep.2023, 13(1):21690.
Front Immunol.2018, 9:2091
Plant Commun.2024, 5(10):101005.
Molecules.2023, 28(9):3685.
Related and Featured Products
Molecules. 2018 Sep 17;23(9). pii: E2367.
Isolation of Novel Sesquiterpeniods and Anti-neuroinflammatory Metabolites from Nardostachys jatamansi.[Pubmed:
30227591]
Nardostachys jatamansi contains various types of sesquiterpenoids that may play an important role in the potency of plant's anti-inflammatory effects, depending on their structure.
METHODS AND RESULTS:
In this study, five new sesquiterpenoids, namely kanshone L (1), kanshone M (2), 7-methoxydesoxo-narchinol (3), kanshone N (4), and nardosdaucanol (5), were isolated along with four known terpenoids (kanshone D (6), nardosinanone G (7), narchinol A (8), and Nardoaristolone B (9)) from the rhizomes and roots of Nardostachys jatamansi. Their structures were determined by analyzing 1D and 2D NMR and MS data. Among the nine sesquiterpenoids, compounds 3, 4, and 8 were shown to possess dose-dependent inhibitory effects against lipopolysaccharide (LPS)-stimulated nitric oxide (NO) production in BV2 microglial cells. Furthermore, compounds 3, 4, and 8 exhibited anti-neuroinflammatory effects by inhibiting the production of pro-inflammatory mediators, including prostaglandin E₂ (PGE₂), inducible nitric oxide synthase (iNOS), and cyclooxygenase-2 (COX-2) proteins, as well as pro-inflammatory cytokines, such as interleukin (IL)-1β, IL-12 and tumor necrosis factor-α (TNF-α), in LPS-stimulated BV2 microglial cells. Moreover, these compounds were shown to inhibit the activation of the NF-κB signaling pathway in LPS-stimulated BV2 microglial cells by suppressing the phosphorylation of IκB-α and blocking NF-κB translocation.
CONCLUSIONS:
In conclusion, five new and four known sesquiterpenoids were isolated from Nardostachys jatamansi, and compounds 3, 4, and 8 exhibited anti-neuroinflammatory effects in LPS-stimulated BV2 microglial cells through inhibiting of NF-κB signaling pathway.
Org Lett. 2014 Aug 15;16(16):4252-5.
Total synthesis of (±)-nardoaristolone B and its analogues.[Pubmed:
25080212]
The first total synthesis of Nardoaristolone B, a nor-sesquiterpenoid with an unusual fused ring system and having protective effects on the injury of neonatal rat cardiomyocytes, has been accomplished.
METHODS AND RESULTS:
Stereoselective synthesis of its novel analogues inlcuding exo-cyclopropyl ring fusion is also part of this disclosure. In addition, an alternate and more efficient one-step method to make a 3/5/6 tricyclic ring system using the Robinson annulation method has been developed toward the generation of a library of compounds around this skeleton.
Org Lett. 2013 Mar 1;15(5):1000-3.
Nardoaristolones A and B, two terpenoids with unusual skeletons from Nardostachys chinensis Batal.[Pubmed:
23394111]
METHODS AND RESULTS:
Nardoaristolone A and Nardoaristolone B, two novel terpenoids derived from the aristolane-type sesquiterpenoid, were isolated from the underground parts of Nardostachys chinensis Batal.
CONCLUSIONS:
Nardoaristolone A is the first reported aristolane-chalcone derivative, while Nardoaristolone B possesses a nor-aristolane sesquiterpenoid skeleton with an unusual 3/5/6 tricyclic ring system. Their structures were elucidated by spectroscopic measurements, and the absolute configurations were established by single-crystal X-ray diffraction experiments.
ACS Omega, 2019, 4(1):2188-2195.
Insect-Repellent and Mosquitocidal Effects of Noreremophilane- and Nardoaristolone-Based Compounds[Reference:
WebLink]
METHODS AND RESULTS:
Here, we disclose novel mosquito-repellentsynthetic hydrindanesbased on noreremophilanes and Nardoaristolone B which show increasedactivity against adult females of Aedes aegypti.The noreremophilanes and Nardoaristolone B with hydrindane skeletonare structurally related to nootkatone with decalin skeleton, a well-studiednatural product extracted from a grape fruit.
CONCLUSIONS:
Out of our library ofcompounds synthesized based on the noreremophilanes and nardoaristoloneB scaffolds, NDS-100598 (compound 20) exhibits higherrepellent and knock-down effects at a very low concentration (0.25mg/cm2), while a few analogues showed considerably enhancedactivity compared to racemic nootkatone. This is the first reportdocumenting insect-repellent and knock-down activity of the noreremophilanesclass of compounds.