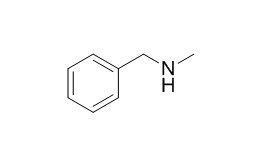
N-Methylbenzylamine
Reference standards.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
BMC Complement Altern Med.2014, 14:242
Nat Commun.2024, 15(1):8221.
Eur J Pharmacol.2021, 899:174010.
Biochem Biophys Res Commun.2018, 505(4):1148-1153
Agronomy2023, 13(6), 1435.
Proc Biol Sci.2024, 291:20232298.
Antioxidants (Basel).2020, 9(6):544.
Microchemical Journal2022, 182: 107874.
Postharvest Biol Tec2019, 149:18-26
Univerzita Karlova2022, 228192.
Related and Featured Products
Chinese Journal of Chemical Physics, 2004, 17(1):61-64.
Photochemical reactions of Hypocrellin A with dibenzylamine and N-methylbenzylamine.[Reference:
WebLink]
METHODS AND RESULTS:
In order to discuss the free radicals formation me ch anism of Hypocrellin A (HA) with amino derivatives, the electron-spin resonance (ESR) spectroscopy was adopted to study the photochemistry on HA with dibenzyl amine (DBA) and
N-Methylbenzylamine (NMBA), respectively. When HA with DBA or NMBA in chloroform solution was illuminated with visible light, singlet oxygen, semiquinone radical and oxynitride radical were formed depending on the conditi on of the solvent system containing the amino-substituted and solved oxygen. Th e signal intensity of oxynitride radical decreased with increasing the illuminat ion time, and the signal intensity of semiquinone radical increased with increas ing the illumination time. The oxynitride radical content was in inverse ratio w ith the semiquinone radical generated by being irradiated.
CONCLUSIONS:
In the aerobic system of chloroform solution containing DBA/HA, smiquinone radical was the main radic al irradiated. The results indicated that HA induced amino derivatives into HA s emiquinone radical.