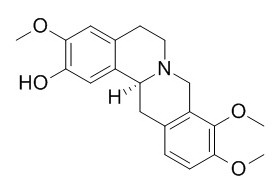
(-)-Isocorypalmine
(-)-Isocorypalmine has significant antifungal activity. l-isocorypalmine acts as a D1 partial agonist and a D2 antagonist to produce its in vivo effects and may be a promising agent for treatment of cocaine addiction.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Mol Plant Pathol.2022, 10.1111:mpp.13280.
Separation Science Plus2022, sscp.202200048.
J Ginseng Res.2023, 47(4):572-582.
bioRxiv2021, 462065.
Int J Mol Sci.2022, 23(11):6104.
J Ethnopharmacol.2017, 209:305-316
Biochem Biophys Res Commun.2019, 518(4):732-738
Molecules.2019, 24(20):3755
Phytomedicine.2021, 84:153501.
Chem Biol Interact.2019, 298:1-7
Related and Featured Products
FEBS Lett . 2014 Jan 3;588(1):198-204.
Cloning and characterization of canadine synthase involved in noscapine biosynthesis in opium poppy[Pubmed:
24316226]
Abstract
Noscapine biosynthesis in opium poppy is thought to occur via N-methylcanadine, which would be produced through 9-O-methylation of (S)-scoulerine, methylenedioxy bridge formation on (S)-tetrahydrocolumbamine, and N-methylation of (S)-canadine. Only scoulerine 9-O-methyltransferase has been functionally characterized. We report the isolation and characterization of a cytochrome P450 (CYP719A21) from opium poppy that converts (S)-tetrahydrocolumbamine to (S)-canadine. Recombinant CYP719A21 displayed strict substrate specificity and high affinity (Km=4.63±0.71 μM) for (S)-tetrahydrocolumbamine. Virus-induced gene silencing of CYP719A21 caused a significant increase in (S)-tetrahydrocolumbamine accumulation and a corresponding decrease in the levels of putative downstream intermediates and noscapine in opium poppy plants.
Keywords: BIA; Benzylisoquinoline alkaloid; CID; CPR; CYP; Cytochrome P450; ESI; GAPDH; LC–MS/MS; Methylenedioxy bridge; Noscapine biosynthesis; Papaver somniferum; SOMT; TNMT; TRV; VIGS; Virus-induced gene silencing; benzylisoquinoline alkaloid; collision-induced dissociation; cytochrome P450; cytochrome P450 reductase; electrospray ionization; glyceraldehyde 3-phosphate dehydrogenase; liquid chromatography–tandem mass spectrometry; scoulerine 9-O-methyltransferase; tetrahydroprotoberberine N-methyltransferase; tobacco rattle virus; virus-induced gene silencing.
Drug Alcohol Depen., 2013, 133(2):693- 703.
L-isocorypalmine reduces behavioral sensitization and rewarding effects of cocaine in mice by acting on dopamine receptors.[Pubmed:
24080315 ]
We previously reported isolation of l-isocorypalmine (l-ICP), a mono-demethylated analog of l-tetrahydropalmatine (l-THP), from the plant Corydalis yanhusuo. Here we characterized its in vitro pharmacological properties and examined its effects on cocaine-induced behaviors in mice.
METHODS AND RESULTS:
Receptor binding, cAMP and [(35)S]GTPγS assays were used to examine pharmacological actions of l-ICP in vitro. Effects of l-ICP on cocaine-induced locomotor hyperactivity and sensitization and conditioned place preference (CPP) in mice were investigated. HPLC was employed to analyze metabolites of l-ICP in mouse serum.
Among more than 40 targets screened, l-ICP and l-THP bound only to dopamine (DA) receptors. l-ICP was a high-affinity partial agonist of D1 and D5 receptors and a moderate-affinity antagonist of D2, D3 and D4 receptors, whereas l-THP bound to only D1 and D5 receptors, with lower affinities than l-ICP. At 10mg/kg (i.p.), l-ICP inhibited spontaneous locomotor activity for a shorter time than l-THP. Pretreatment with l-ICP reduced cocaine-induced locomotor hyperactivities. Administration of l-ICP before cocaine once a day for 5 days reduced cocaine-induced locomotor sensitization on days 5 and 13 after 7 days of withdrawal. Pretreatment with l-ICP before cocaine daily for 6 days blocked cocaine-induced CPP, while l-ICP itself did not cause preference or aversion. HPLC analysis showed that l-ICP was the main compound in mouse serum following i.p. injection of l-ICP.
CONCLUSIONS:
l-ICP likely acts as a D1 partial agonist and a D2 antagonist to produce its in vivo effects and may be a promising agent for treatment of cocaine addiction.