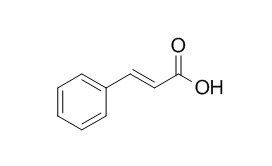
Cinnamic acid
Cinnamic acid, a naturally occurring aromatic fatty acid of low toxicity, has anti-diabetic ,
anticancer and antioxidant activities. It is a cell differentiation inducer and protein isoprenylation inhibitor, shows a significant radio-protective effect by reducing the DNA damage induced by X-rays. Cinnamic acid inhibited feeding by detritivores, this inhibition occurs at concentrations found in nature and may be a major factor controlling the rate of decay of organic matter. It inhibited mushroom tyrosinase activity in reversiblywith the IC 50 value of 2.10 mM.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Mol Pharm.2018, 15(8):3285-3296
Environ Toxicol.2023, 38(7):1641-1650.
Mol Med Rep.2022, 25(1):8.
Cardiovasc Toxicol.2021, 21(11):947-963.
Molecules.2023, 28(19):6767.
LWT2020, 126:109313
Sci Rep.2017, 7:467-479
Expert Opin Ther Targets.2024, :1-11.
Vietnam Journal of Science2022, 64(2), 69-75.
Journal of Cluster Science2024, 35:635-656.
Related and Featured Products
Mutat Res Genet Toxicol Environ Mutagen. 2014 Aug;770:72-9.
Radio-protective effect of cinnamic acid, a phenolic phytochemical, on genomic instability induced by X-rays in human blood lymphocytes in vitro.[Pubmed:
25344167]
METHODS AND RESULTS:
The present study was designed to determine the protective activity of Cinnamic acid against induction by X-rays of genomic instability in normal human blood lymphocytes. This radio-protective activity was assessed by use of the cytokinesis-block micronucleus test and the alkaline comet assay, with human blood lymphocytes isolated from two healthy donors. A Siemens Mevatron MD2 (Siemens AG, USA, 1994) linear accelerator was used for the irradiation with 1 or 2 Gy. Treatment of the lymphocytes with Cinnamic acid prior to irradiation reduced the number of micronuclei when compared with that in control samples. Treatment with Cinnamic acid without irradiation did not increase the number of micronuclei and did not show a cytostatic effect in the lymphocytes. The results of the alkaline comet assay revealed that Cinnamic acid reduces the DNA damage induced by X-rays, showing a significant radio-protective effect. Cinnamic acid decreased the frequency of irradiation-induced micronuclei by 16-55% and reduced DNA breakage by 17-50%, as determined by the alkaline comet assay.
CONCLUSIONS:
Cinnamic acid may thus act as a radio-protective compound, and future studies may focus on elucidating the mechanism by which Cinnamic acid offers radioprotection.
Plant & Soil, 2004, 263(1):143-50.
Incidence of Fusarium wilt in Cucumis sativus L. is promoted by cinnamic acid, an autotoxin in root exudates[Reference:
WebLink]
METHODS AND RESULTS:
The effects of Fusarium oxysporum f. sp. cucumerinum, the pathogen causing Fusarium wilt in cucumber and Cinnamic acid, a principal autotoxic component in the root exudates of cucumber (Cucumis sativus L.), on plant growth, Photosynthesis and incidence of Fusarium wilt in cucumber were studied in order to elucidate the interaction of autotoxins and soil-borne pathogens in the soil sickness. F. oxysporum. f. sp. cucumerinum (FO) and Cinnamic acid (CA) at 0.1 or 0.25 mM significantly decreased net photosynthetic rate, stomatal conductance and the quantum yield of Photosystem II photochemistry (ΦPSII), followed by a reduction of plant biomass production, but did not induce photoinhibition. Pretreatment with CA before inoculation with FO increased the effectiveness of FO, together with a slight photoinhibition. CA pretreatment significantly increased percentage of plants affected by Fusarium wilt, browning index of vascular bundle and Fusarium population in the nutrient solution.
CONCLUSIONS:
All these results indicate that CA enhanced Fusarium wilt by predisposing cucumber roots to infection by FO through a direct biochemical and physiological effect. It is likely that soil sickness results from an interaction of many factors.
Nature, 1979, 280(280):55-7.
Cinnamic acid inhibition of detritus feeding. [Reference:
WebLink]
AN important advance in ecology has been the general acceptance of Fraenkel's postulate that certain chemicals in plants deter herbivores1–3. Such chemicals are usually termed secondary plant substances because they are not involved in primary metabolic pathways. Very little of the annual production of biomass by higher plants is consumed by herbivores or phyto-pathogens4. Instead, most of the biomass becomes litter and eventually decays through the activity of decomposers. The secondary compounds that deter grazers while the plants are alive do not disappear immediately when plants senesce and die. We have therefore investigated whether these anti-herbivore substances continue to inhibit consumption by organisms feeding on litter or detritus. We report here that Cinnamic acids, one type of secondary plant substances found in detritus, inhibit feeding by detritivores. This inhibition occurs at concentrations found in nature and may be a major factor controlling the rate of decay of organic matter.
2018 Sep 4.
Antimicrobial activity of trans-cinnamic acid and commonly used antibiotics against important fish pathogens and nonpathogenic isolates[Pubmed:
30179290]
Aims: Antibiotics and several other chemicals have been used to prevent fish diseases. However, this situation results in economic loss for the companies in the aquaculture industry and most importantly it pollutes the environment. Cinnamic acid is a naturally occurring aromatic acid and is considered to be safe for human consumption. Therefore, in this study, the antibacterial activity of trans-Cinnamic acid and commonly used antibiotics, namely chloramphenicol, vancomycin, streptomycin and erythromycin, were tested against 32 bacteria, including fish pathogens, nonpathogenic isolates and collection strains.
Methods and results: Trans-Cinnamic acid was applied against the bacteria using the disc diffusion and microdilution method under in vitro conditions. Antibiotics were also tested under similar conditions against all tested bacteria using the disc diffusion method. The results show that among 32 bacterial strains trans-Cinnamic acid exhibited potent inhibitory effect on the Gram-negative fish pathogen Aeromonas sobria. In addition, a moderate inhibition of trans-Cinnamic acid of fish pathogens Aeromonas salmonicida, Vibrio (Listonella) anguillarum, Vibrio crassostreae and Yersinia ruckeri was also observed for trans-Cinnamic acid in our study. On the contrary, the majority of nonpathogenic intestinal isolates were resistant to trans-Cinnamic acid.
Conclusions: To the best of our knowledge, this is the first report on the antimicrobial activity of trans-Cinnamic acid on 24 of the studied bacteria isolated from fish. In conclusion, trans-Cinnamic acid can be used as an environmentally friendly alternative additive to prevent and control primarily A. sobria, as well as other pathogenic bacteria such as A. salmonicida, V. anguillarum, V. crassostreae and Y. ruckeri.
Significance and impact of the study: This study indicated that trans-Cinnamic acid may present an environmentally friendly alternative therapeutic agent against A. sobria infections in the aquaculture industry.
Keywords: antimicrobial; fish pathogens; intestine; phenolic organic acid; trans-Cinnamic acid.
Phytomedicine. 2015 Feb 15;22(2):297-300.
Cinnamic acid exerts anti-diabetic activity by improving glucose tolerance in vivo and by stimulating insulin secretion in vitro.[Pubmed:
25765836]
Although the anti-diabetic activity of Cinnamic acid, a pure compound from cinnamon, has been reported but its mechanism(s) is not yet clear.
METHODS AND RESULTS:
The present study was designed to explore the possible mechanism(s) of anti-diabetic activity of Cinnamic acid in in vitro and in vivo non-obese type 2 diabetic rats. Non-obese type 2 diabetes was developed by injecting 90 mg/kg streptozotocin in 2-day-old Wistar pups. Cinnamic acid and cinnamaldehyde were administered orally to diabetic rats for assessing acute blood glucose lowering effect and improvement of glucose tolerance. Additionally, insulin secretory activity of Cinnamic acid and cinnamaldehyde was evaluated in isolated mice islets. Cinnamic acid, but not cinnamaldehyde, decreased blood glucose levels in diabetic rats in a time- and dose-dependent manner. Oral administration of Cinnamic acid with 5 and 10 mg/kg doses to diabetic rats improved glucose tolerance in a dose-dependent manner. The improvement by 10 mg/kg Cinnamic acid was comparable to that of standard drug glibenclamide (5 mg/kg). Further in vitro studies showed that cinnamaldehyde has little or no effect on glucose-stimulated insulin secretion; however, Cinnamic acid significantly enhanced glucose-stimulated insulin secretion in isolated islets.
CONCLUSIONS:
In conclusion, it can be said that Cinnamic acid exerts anti-diabetic activity by improving glucose tolerance in vivo and stimulating insulin secretion in vitro.
Int J Biochem Cell Biol. 2008;40(9):1918-29.
Anticancer effects of ginsenoside Rg1, cinnamic acid, and tanshinone IIA in osteosarcoma MG-63 cells: nuclear matrix downregulation and cytoplasmic trafficking of nucleophosmin.[Pubmed:
18403247]
Ginsenoside Rg1, Cinnamic acid, and tanshinone IIA are effective anticancer and antioxidant constituents of traditional Chinese herbal medicines of Ginseng (Panax ginseng), Xuanshen (Radix scrophulariae), and Danshen (Salvia mitiorrhiza), respectively. There was insufficient study on molecular mechanisms of anticancer effects of those constituents and their targets were unknown.
METHODS AND RESULTS:
We chose nucleophosmin as a candidate molecular target because it is frequently mutated and upregulated in various cancer cells. Nucleophosmin is a major nucleolus phosphoprotein that involves in rRNA synthesis, maintaining genomic stability, and normal cell division and its haploinsufficiency makes cell more susceptible to oncogenic assault. Ginsenoside Rg1, Cinnamic acid, and tanshinone IIA treatment of osteosarcoma MG-63 cells decreased nucleophosmin expression in nuclear matrix and induced nucleophosmin translocation from nucleolus to nucleoplasm and cytoplasm, a process of dedifferentiating transformed cells. Using immunogold electro-microscopy, we found at the first time that nucleophosmin was localized on nuclear matrix intermediate filaments that had undergone restorational changes after the treatments. Nucleophosmin also functions as a molecular chaperone that might interact with multiple oncogenes and tumor suppressor genes. We found that oncogenes c-myc, c-fos and tumor suppressor genes, P53, Rb were regulated by ginsenoside Rg1, Cinnamic acid, and tanshinone IIA as well. In present study, we identified nucleophosmin as a molecular target of the effective anticancer constituents of t Ginseng, Xuanseng, and Danseng that down-regulated nucleophosmin in nuclear matrix, changed its trafficking from nucleolus to cytoplasm, and regulated several oncogenes and tumor suppressor genes.
CONCLUSIONS:
Therefore, we postulate that Ginsenoside Rg1, Cinnamic acid, and tanshinone IIA could serve as protective agents in cancer prevention and treatment.
Food Chem., 2005, 92(4):707-12.
Inhibitory effects of cinnamic acid and its derivatives on the diphenolase activity of mushroom (Agaricus bisporus) tyrosinase[Reference:
WebLink]
The effects of Cinnamic acid and its derivatives (2-hydroxyCinnamic acid, 4-hydroxyCinnamic acid and 4-methoxyCinnamic acid) on the activity of mushroom tyrosinase have been studied. Results showed that Cinnamic acid, 4-hydroxyCinnamic acid and 4-methoxyCinnamic acid strongly inhibited the diphenolase activity of mushroom tyrosinase and the inhibition was reversible. The IC50 values were estimated to be 2.10, 0.50 and 0.42 mM, respectively. 2-HydroxyCinnamic acid had no inhibitory effect on the diphenolase activity of the enzyme. Kinetic analyses showed that the inhibition type of Cinnamic acid and 4-methoxyCinnamic acid was noncompetitive with the constants (KI) determined to be 1.994 and 0.458 mM, respectively. The inhibition type of 4-hydroxyCinnamic acid was competitive, with the inhibition constant (KI) was 0.244 mM.