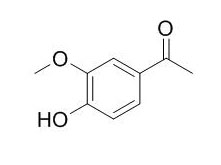
Acetovanillone
Acetovanillone is the NADPH oxidase inhibitor, induces oxidative stress.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Molecules.2024, 29(22):5260.
Biomed Pharmacother.2022, 145:112474.
Foods.2024, 13(23):3861.
Nutrients.2021, 13(12):4364.
The Catharanthus Genome2022,35-83.
Metabolites.2020, 10(11):440.
Biomed Pharmacother.2024, 175:116770.
Prev Nutr Food Sci.2024, 29(4):563-571.
Emirates Journal of Food and Agriculture.2022, 34(6): 528-536.
Food Chem Toxicol.2023, 176:113802.
Related and Featured Products
Toxicol Appl Pharmacol. 2006 May 1;212(3):179-87. Epub 2005 Aug 24.
The NADPH oxidase inhibitor apocynin (acetovanillone) induces oxidative stress.[Pubmed:
16120450]
Apocynin (Acetovanillone) is often used as a specific inhibitor of NADPH oxidase.
METHODS AND RESULTS:
In N11 glial cells, apocynin induced, in a dose-dependent way, a significant increase of both malonyldialdehyde level (index of lipid peroxidation) and lactate dehydrogenase release (index of a cytotoxic effect). Apocynin evoked also, in a significant way, an increase of H(2)O(2) concentration and a decrease of the intracellular glutathione/glutathione disulfide ratio, accompanied by augmented efflux of glutathione and glutathione disulfide. Apocynin induced the activation of both pentose phosphate pathway and tricarboxylic acid cycle, which was blocked when the cells were incubated with glutathione together with apocynin. The cell incubation with glutathione prevented also the apocynin-induced increase of malonyldialdehyde generation and lactate dehydrogenase leakage. Apocynin exerted an oxidant effect also in a cell-free system: indeed, in aqueous solution, it evoked a faster oxidation of the thiols glutathione and dithiothreitol, and elicited the generation of reactive oxygen species, mainly superoxide anions.
CONCLUSIONS:
Our results suggest that apocynin per se can induce an oxidative stress and exert a cytotoxic effect in N11 cells and other cell types, and that some effects of apocynin in in vitro and in vivo experimental models should be interpreted with caution.
Applied Microbiology & Biotechnology, 2008 , 81 (4) :783-90.
Synergistic effect of laccase mediators on pentachlorophenol removal by Ganoderma lucidum laccase.[Pubmed:
18987855]
Laccases have low redox potentials limiting their environmental and industrial applications. The use of laccase mediators has proven to be an effective approach for overcoming the low redox potentials. However, knowledge about the role played by the mediator cocktails in such a laccase-mediator system (LMS) is scarce.
METHODS AND RESULTS:
Here, we assembled different dual-agent mediator cocktails containing 2,2'-azino-bis-(3-ethylbenzothiazoline-6-sulfonate) (ABTS), vanillin, and/or Acetovanillone, and compared their mediating capabilities with those of each individual mediator alone in oxidation of pentachlorophenol (PCP) by Ganoderma lucidum laccase. Cocktails containing ABTS and either vanillin or Acetovanillone strongly promoted PCP removal compared to the use of each mediator alone. The removal enhancement was correlated with mediator molar ratios of the cocktails and incubation times. Analysis of the kinetic constants for each mediator compound showed that G. lucidum laccase was very prone to react with ABTS rather than vanillin and Acetovanillone in the cocktails. Moreover, the presence of the ABTS radical (ABTS+*) and vanillin or Acetovanillone significantly enhanced PCP removal concomitant with electron transfer from vanillin or Acetovanillone to ABTS+*.
CONCLUSIONS:
These results strongly suggest that vanillin and Acetovanillone mediate the reaction between ABTS and PCP via multiple sequential electron transfers among laccase and its mediators.