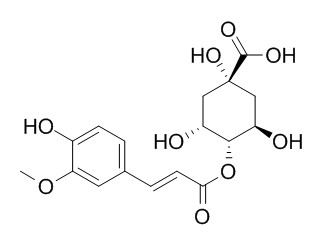
4-O-Feruloylquinic acid
4-O-Feruloylquinic acid can be useful for the prevention of cardiovascular and other chronic diseases.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Research Square2022, rs.3.rs-1948239
FASEB J.2019, 33(2):2026-2036
Cell Prolif.2021, 54(8):e13083.
J Ginseng Res.2020, 44(4):611-618.
AMB Express2020. 10(1):126.
Pharmaceutics2022, 14(2),376.
Nutrients.2023, 15(4):954.
The Journal of Supercritical Fluids2021, 176:105305.
Separations2023, 10(4),255.
Naunyn Schmiedebergs Arch Pharmacol.2017, 390(10):1073-1083
Related and Featured Products
Phytochem Anal. 2007 May-Jun;18(3):213-8.
Isolation of chlorogenic acids and their derivatives from Stemona japonica by preparative HPLC and evaluation of their anti-AIV (H5N1) activity in vitro.[Pubmed:
17500364]
Two chlorogenic acids and five chlorogenic acid derivatives were simultaneously separated and purified from Stemona japonica by preparative high-performance liquid chromatography.
METHODS AND RESULTS:
Five of the collected compounds were over 95% pure while the other two compounds were over 90% pure. Their structures were elucidated as 3-O-feruloylquinic acid (1), 4-O-Feruloylquinic acid (2), methyl 3-O-feruloylquinate (3), methyl 5-O-caffeyolquinate (4), methyl 4-O-feruloylquinate (5), ethyl 3-O-feruloylquinate (6) and the new compound ethyl 4-O-feruloylquinate (7) by UV, NMR and ESI-MS. All compounds were obtained from Stemona species for the first time, however compounds 6 and 7 are believed to be artefacts from the ethanol extraction. The anti-AIV (H5N1) activities were evaluated by Neutral Red uptake assay.
CONCLUSIONS:
Compounds 3 and 4 exerted moderate inhibitory effect against AIV (H5N1) in vitro.