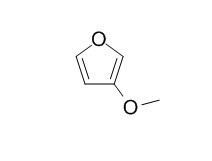
3-Methoxyfuran
3-Methoxyfuran shows Diels-Alder reactivity under forcing conditions.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Molecules.2016, 21(10)
bioRxiv2021, 458409.
Molecules.2023, 28(13):4972.
Biosci Biotechnol Biochem.2020, 84(3):621-632
Exp Neurobiol.2018, 27(3):200-209
Molecules.2020 ,25(16):3697.
Molecules.2019, 24(7):E1290
Biomedicines.2022, 10(5):1170
Manomaniam Sundaranar University2023, 3859769.
Nutrients.2023, 15(3):753.
Related and Featured Products
J Phys Chem A. 2010 Aug 5;114(30):7984-95.
Structure and thermochemical properties of 2-methoxyfuran, 3-methoxyfuran, and their carbon-centered radicals using computational chemistry.[Pubmed:
20666545]
METHODS AND RESULTS:
Methoxyfurans are known components in a number of biofuel synthesis processes and their thermochemical properties are important to the stability, reaction paths, and chemical kinetics of these species. Enthalpies (DeltaH degrees (f298)), entropies (S degrees (298)), and heat capacities (C(p)(T)) are reported for 2-methoxyfuran and 3-Methoxyfuran, cyclic ethers with possible biofuel implications, and their radicals corresponding to loss of hydrogen atoms. Standard enthalpies of formation are calculated at the B3LYP/6-31G(d,p), B3LYP/6-311G(2d,2p), CBS-QB3, G3MP2B3, and G3 levels of theory with isodesmic reactions to minimize calculation errors. Structures, vibrational frequencies, and internal rotor potentials are calculated at the B3LYP/6-31G(d,p) density functional level and are used to determine the entropy and heat capacities. The recommended ideal gas phase enthalpy of formation, from the average of the CBS-QB3 and G3MP2B3 levels of theory, for 2-methoxyfuran is -45.0 kcal mol(-1) and for 3-Methoxyfuran is -41.1 kcal mol(-1). Bond dissociation energies are also calculated. The C-H bonds of the furan ring are approximately 120 kcal mol(-1), which is consistent with recent data on several alkylfurans; they are significantly stronger than non-aromatic, stable heterocyclic structures.
CONCLUSIONS:
The bond energy decreases to 98 kcal mol(-1) for the methoxy-methyl C-H bonds making this methyl site a favorable abstraction target and an important site for initial decomposition paths during combustion. Group additivity for furan is discussed and groups for furan and methoxyfuran carbon radicals are derived.