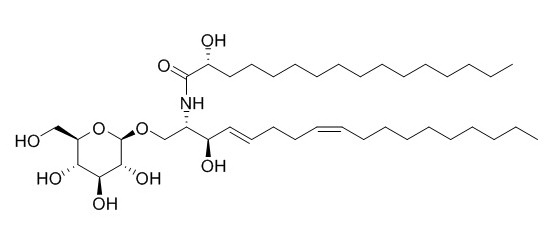
Soyacerebroside II
Soyacerebroside II exhibits ionophoretic activity for Ca2+ ion. Soyacerebrosides I and II have modulating the cellular immune response effects, they show obvious inhibitory activity on IL-18 secretion in human peripheral blood mononuclear cells (PBMC).
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Biomolecules.2024, 14(4):451.
Sains Malaysiana2024, 53(4):795-805
Pharmaceuticals (Basel).2020, 13(10):302.
Molecules 2022, 27(3),960.
J Cell Biochem.2024, 125(4):e30537.
Front Endocrinol (Lausanne).2023, 14:1138676.
Sci Rep.2023, 13(1):13072.
Planta Med.2022, a-1876-3009.
Antioxidants (Basel).2021, 10(10):1620.
Wageningen University & Research2018, January 2018
Related and Featured Products
Chem Pharm Bull (Tokyo). 1990 Nov;38(11):2933-8.
Sphingolipids and glycerolipids. I. Chemical structures and ionophoretic activities of soya-cerebrosides I and II from soybean.[Pubmed:
2085876]
METHODS AND RESULTS:
Two glycosphingolipids named soya-cerebrosides I and II were isolated from soybean, the seeds of Glycine max Merrill (Leguminosae), and their chemical structures have been elucidated on the basis of physicochemical evidence and several chemical degradation reactions.
CONCLUSIONS:
By using a newly constructed liquid membrane-type apparatus (W-08) for measurement of ion-transport and ion-binding activities and by employing a method using human erythrocyte membranes for measurement of ion-permeability, it has been found that Soyacerebroside II exhibits ionophoretic activity for Ca2+ ion.
Lipids. 2009 Aug;44(8):759-63.
An isomeric mixture of novel cerebrosides isolated from Impatiens pritzellii reduces lipopolysaccharide-induced release of IL-18 from human peripheral blood mononuclear cells.[Pubmed:
19609788]
An isomeric mixture of two cerebrosides, soyacerebroside I and Soyacerebroside II, was isolated from an ethno drug, the rhizomes of Impatiens pritzellii Hook. f. var. hupehensis Hook. f., and their structures were identified by spectroscopic (NMR, MS) analysis.
METHODS AND RESULTS:
In order to determine the immunomodulatory activities of soya-cerebrosides I and II, the effects of the mixture of cerebrosides (MC) on cytotoxicity of human peripheral blood mononuclear cells (PBMC) and the inhibitory activities to lipopolysaccharide (LPS)-induced interleukin (IL)-18 in PBMC were studied. The MC at concentrations of 10 and 1 microM, without toxicity to PBMC in 24 h, showed obvious inhibitory activity on IL-18 secretion.
CONCLUSIONS:
Because of this effect of modulating the cellular immune response, soya-cerebrosides I and II were considered to be the active substances of this ethno drug.
Zhongguo Zhong Yao Za Zhi. 2014 Jan;39(2):258-61.
Chemical constituents from leaves of Ilex latifolia.[Pubmed:
24761642]
METHODS AND RESULTS:
Nine compounds were isolated from the leaves of Ilex latifolia. Their structures were respectively identified as 5-hydroxy-6, 7, 8, 4'-tetramethoxyflavone (1), tangeretin (2), nobiletin (3), 5-hydroxy-6, 7, 8, 3', 4'-pentamethoxyflavone (4), 5, 6, 7, 8, 4'-pentamethoxyflavonol (5), 5, 6, 7, 8, 3', 4'-hexamethoxy-flavonol (6), 5-hydroxy-3', 4', 7-trimethoxyflavanone (7), soyacerebroside I (8), and Soyacerebroside II (9) by their physicochemical properties and spectroscopic data.
CONCLUSIONS:
Compounds 1-9 were isolated from this plant for the first time.