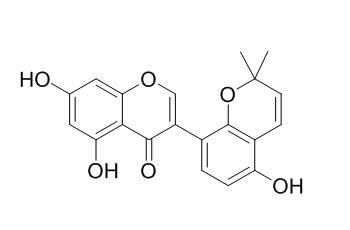
Sophoraisoflavone A
Sophoraisoflavone A is a potential MRP inhibitor; it is also an inhibitor of germ tube growth in the AM fungus Gigaspora margarita, it strongly inhibited germ tube growth at 1.25 ug/disc. Sophoraisoflavone A shows inhibitory effects on copper-induced protein oxidative modification of mice brain homogenate in vitro.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Mediators Inflamm.2016, 2016:7216912
Univerzita Karlova2021, 20.500.11956.
Phytother Res.2019, 33(3):676-689
Molecules.2024, 29(3):671.
ACS Omega.2021, 6(36):23460-23474.
Front Microbiol.2024, 15:1429027.
J Sci Food Agric.2018, 98(3):1153-1161
Int J Mol Sci.2023, 24(24):17589.
Food Funct.2022, 13(23):12105-12120.
Sci Rep.2019, 9(1):18080
Related and Featured Products
Phytochemistry. 2010 Nov;71(16):1865-71.
Lupin pyranoisoflavones inhibiting hyphal development in arbuscular mycorrhizal fungi.[Pubmed:
20813384]
METHODS AND RESULTS:
White lupin (Lupinus albus L.), a non-host plant for arbuscular mycorrhizal (AM) fungi in the typically mycotrophic family Fabaceae, has been investigated for root metabolites that inhibit hyphal development in AM fungi. Four known pyranoisoflavones, licoisoflavone B (1), Sophoraisoflavone A (2), alpinumisoflavone (3) and 3'-hydroxy-4'-O-methylalpinumisoflavone (4), together with three previously unknown pyranoisoflavones, lupindipyranoisoflavone A (5), 10'-hydroxylicoisoflavone B (6) and 10'-hydroxySophoraisoflavone A (7) were isolated from the root exudates of white lupin as an inhibitor of germ tube growth in the AM fungus Gigaspora margarita.
CONCLUSIONS:
Pyranoisoflavones 1, 2 and 3 strongly inhibited germ tube growth at 0.63, 1.25 and 0.63 μg/disc, respectively. The remaining compounds 4-7 were either moderate or weak inhibitors that inhibited germ tube growth at concentrations higher than 10 μg/disc. Licoisoflavone B (1) and Sophoraisoflavone A (2) completely inhibited hyphal branching induced by a lupin strigolactone, orobanchyl acetate, in G. margarita at 0.16 and 0.63 μg/disc, respectively.
Biol Trace Elem Res. 2001 Aug;81(2):169-75.
Inhibitory effects of licoisoflavones A and B and sophoraisoflavone A of Sophra mooracroftiana Beth ex Baker on copper-ion-induced protein oxidative modification of mice brain homogenate, in vitro.[Pubmed:
11554397 ]
METHODS AND RESULTS:
We present the results of an in vitro investigation of the inhibitory effects of licoisoflavones A and B and Sophoraisoflavone A isolated from Sophra mooracroftiana BETH ex BAKER on copper-induced protein oxidative modification of mice brain homogenate in vitro. Although inhibitory effect of Sophoraisoflavone A was stronger than those of licoisoflavones A and B, genistein as a related isoflavone, and mannitol as a hydroxy radical scavenger, inhibitory effects of licoisoflavones A and B were weaker than those of genistein and mannitol.
CONCLUSIONS:
These results demonstrated that the difference of inhibitory effects are dependent on the relation between chemical structures of these isoflavones, such as hydroxy group or benzopyran, and oxidative stress.
Blood Cells Mol Dis. 2001 Sep-Oct;27(5):894-900.
Monitoring of MRP-like activity in human erythrocytes: inhibitory effect of isoflavones.[Pubmed:
11783953 ]
METHODS AND RESULTS:
A method to fluorometrically monitor efflux of 2',7'-bis-(carboxypropyl)-5(6)-carboxyfluorescein (BCPCF) from human erythrocytes was developed. Genistein, daidzein, Sophoraisoflavone A, and licoisoflavone A induced 50% inhibition (IC(50)) of BCPCF efflux at 15-70 microM. The IC(50) value of the most efficient isoflavone, licoisoflavone A (15-25 microM), was comparable to that of indomethacin (approximately 10 microM) and markedly lower than for probenecid (100-200 microM), both known MRP1 inhibitors.
CONCLUSIONS:
Our results indicate that the human erythrocyte is a useful cell model in screening potential MRP inhibitors, that BCPCF is a good substrate for MRP, and that some isoflavones at low concentrations inhibit MRP-mediated efflux.