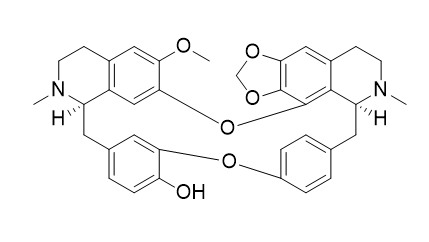
Cepharanoline
Cepharanoline has significant antiplasmodial activity and Proliferative activity of mouse skin cultured hair cells.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Korean J. Medicinal Crop Sci.2022, 30(2):117-123.
Tropical J. of Pha. Research2017, 16(3):543-552
Int J Mol Sci.2019, 20(3):E651
J Sep Sci.2018, 41(9):1938-1946
Revista Brasileira de Farmacognosia2024, 34:1091-1100.
Applied Biological Chemistry2023, 66(58):112.
Nutrients.2024, 16(20):3521.
J Pain Res.2022, 15:3469-3478.
J Cell Mol Med.2023, 27(11):1592-1602.
European Journal of Integrative Medicine2018, 20:165-172
Related and Featured Products
Nat Prod Res . 2010 Nov;24(18):1766-1770.
Antiplasmodial activity of three bisbenzylisoquinoline alkaloids from the tuber of Stephania rotunda[Pubmed:
20981617]
Three bisbenzylisoquinoline alkaloids were isolated for the first time from Stephania rotunda tuber. Their structures were elucidated by spectroscopic methods and their antiplasmodial activity was investigated in vitro on chloroquine resistant Plasmodium falciparum strain W2. These alkaloids were identified as 2-norcepharanthine (1), Cepharanoline (2) and fangchinoline (3). In vitro, they displayed significant antiplasmodial activity with inhibitory concentration 50 values of 0.3, 0.2 and 0.3 μM.
Biochem Pharmacol . 2003 Aug 1;66(3):379-385.
Inhibition of Na(+),K(+)-ATPase by the extract of Stephania cephararantha HAYATA and bisbenzylisoquinoline alkaloid cycleanine, a major constituent[Pubmed:
12907236]
The Stephania cephararantha HAYATA extract, and its constituent bisbenzylisoquinoline alkaloids, such as cycleanine, cepharanthine, isotetrandrine, berbamine, homoaromoline, and Cepharanoline were studied for effects on Na(+),K(+)-ATPase activity. The S. cephararantha HAYATA extract inhibited Na(+),K(+)-ATPase activity with an apparent IC(50) value of 540 microg/mL. Cycleanine markedly inhibited Na(+),K(+)-ATPase activity with an IC(50) value of 6.2 x 10(-4)M. It slightly inhibited Mg(2+)-ATPase, H(+)-ATPase, and Ca(2+)-ATPase. No effects on alkaline and acid phosphatase activities were observed. The inhibition by isotetrandrine, homoaromoline, cepharanthine, and berbamine was less marked, and Cepharanoline showed no effect. Five synthetic analogues of cepharanthine slightly inhibited the activity. The mechanism of inhibition by cycleanine on Na(+),K(+)-ATPase activity was examined in detail, and the following results were obtained in the overall reaction: (1) the mode of inhibition was noncompetitive with respect to ATP; (2) the degree of inhibition was decreased with a decrease of K(+) concentration; (3) it was not affected by Na(+) concentration; (4) the inhibition mechanism was different from that of ouabain. The activity of K(+)-dependent p-nitrophenyl phosphatase, a partial reaction of Na(+),K(+)-ATPase, did not appear to have been inhibited by cycleanine in the reaction mixture containing 15 mM K(+) (optimum condition). However, cycleanine increased the K(0.5) value for K(+) and reduced the K(i) values for Na(+) and ATP, in K(+)-dependent p-nitrophenyl phosphatase. Cycleanine might interact with the enzyme in Na.E(1)-P form and prevents the reaction step from Na.E(1)-P to E(2)-P.
Planta Med . 1997 Oct;63(5):425-428.
Bisbenzylisoquinoline alkaloids from Stephania cepharantha and their effects on proliferation of cultured cells from the murine hair apparatus[Pubmed:
9342946]
Bisbenzylisoquinoline alkaloids were isolated from Stephania cepharantha Hayata and their proliferative activities on cultured hair cells from the murine skin were evaluated. Cepharanthine (1), Cepharanoline (9), isotetrandrine (2), and berbamine (7) showed significant activities in the range of 0.01-0.1 microgram/ml, but had no activity on cultured keratinocytes or fibroblasts from the murine skin.
Nat Prod Res . 2010 Nov;24(18):1766-1770.
Antiplasmodial activity of three bisbenzylisoquinoline alkaloids from the tuber of Stephania rotunda[Pubmed:
20981617]
Three bisbenzylisoquinoline alkaloids were isolated for the first time from Stephania rotunda tuber. Their structures were elucidated by spectroscopic methods and their antiplasmodial activity was investigated in vitro on chloroquine resistant Plasmodium falciparum strain W2. These alkaloids were identified as 2-norcepharanthine (1), Cepharanoline (2) and fangchinoline (3). In vitro, they displayed significant antiplasmodial activity with inhibitory concentration 50 values of 0.3, 0.2 and 0.3 μM.