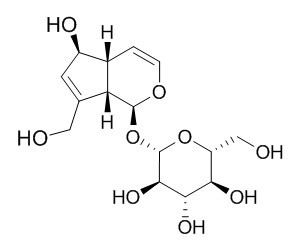
Aucubin
Aucubin is an iridoid glycoside with a wide range of biological activities, including pancreas-protective, chondroprotective, antispasmodic, liver-protective, anti-inflammatory, anti-microbial, antioxidant, anti-algesic as well as anti-tumor activities. Aucubin prevents neuronal death in the hippocampal CA1 region in rats with diabetic encephalopathy, it also has protective effects on H2O2-induced apoptosis in PC12 cells. Aucubin may improve obesity-induced atherosclerosis by attenuating TNF-α-induced inflammatory responses. Aucubin suppresses hepatitis B viral DNA replication in vitro.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
J Ethnopharmacol.2017, 197:157-164
Cancers (Basel).2021, 13(9):2223.
Pest Manag Sci.2023, 79(8):2675-2685.
Jour. of Stored Pro & Postharvest Res.2016, 7(3):32-36
Food Chem.2020, 313:126079
Vietnam Journal of Food Control.2022, 5(2): 115-128.
Integr Med Res.2024, 13(1):101025.
Drug Invention Today2019, 12(6):1303-1306
Clin Exp Pharmacol Physiol.2020, doi: 10.1111
Exp Mol Med.2020, 52(4):629-642.
Related and Featured Products
Exp Neurobiol. 2014 Sep;23(3):238-45.
Aucubin promotes neurite outgrowth in neural stem cells and axonal regeneration in sciatic nerves.[Pubmed:
25258571]
Aucubin is an iridoid glycoside with a wide range of biological activities, including anti-inflammatory, anti-microbial, anti-algesic as well as anti-tumor activities. Recently, it has been shown that Aucubin prevents neuronal death in the hippocampal CA1 region in rats with diabetic encephalopathy. In addition, it has protective effects on H2O2-induced apoptosis in PC12 cells.
METHODS AND RESULTS:
We have shown here that Aucubin promotes neuronal differentiation and neurite outgrowth in neural stem cells cultured primarily from the rat embryonic hippocampus. We also investigated whether Aucubin facilitates axonal elongation in the injured peripheral nervous system. Aucubin promoted lengthening and thickness of axons and re-myelination at 3 weeks after sciatic nerve injury.
CONCLUSIONS:
These results indicate that administration of Aucubin improved nerve regeneration in the rat model of sciatic nerve injury, suggesting that Aucubin may be a useful therapeutic compound for the human peripheral nervous system after various nerve injuries.
Cytokine. 2013 Jun;62(3):407-12.
Aucubin, a naturally occurring iridoid glycoside inhibits TNF-α-induced inflammatory responses through suppression of NF-κB activation in 3T3-L1 adipocytes.[Pubmed:
23612013]
Obesity is closely associated with a state of chronic, low-grade inflammation characterized by abnormal cytokine production and activation of inflammatory signaling pathways in adipose tissue.
Tumor necrosis factor (TNF)-α is chronically elevated in adipose tissues of obese rodents and humans. Increased levels of TNF-α are implicated in the induction of atherogenic adipokines, such as plasminogen activator inhibitor (PAI)-1, adipose-tissue-derived monocyte chemoattractant protein (MCP)-1, and interleukin (IL)-6. Aucubin, an iridoid glycoside existing in medicinal plants, has been reported to show an anti-inflammatory activity by suppression of TNF-α production in murine macrophages.
METHODS AND RESULTS:
The present study is aimed to investigate the effects of Aucubin on TNF-α-induced atherogenic changes of the adipokines in differentiated 3T3-L1 cells. Aucubin significantly inhibited TNF-α-induced secretion and mRNA synthesis of the atherogenic adipokines including PAI-1, MCP-1, and IL-6. Further investigation of the molecular mechanism revealed that pretreatment with Aucubin suppressed extracellular signal-regulated kinase (ERK) activation, inhibitory kappa Bα (IκBα) degradation, and subsequent nuclear factor kappa B (NF-κB) activation.
CONCLUSIONS:
These findings suggest that Aucubin may improve obesity-induced atherosclerosis by attenuating TNF-α-induced inflammatory responses.
Res Commun Mol Pathol Pharmacol. 1998 Nov;102(2):189-204.
Liver-protective activities of aucubin derived from traditional oriental medicine.[Pubmed:
10100510]
The iridoid glycosides including Aucubin (AU), catalpol (CA), swertimarin (SW), and gardenoside (GA) are frequently found as natural constituents of many traditional oriental medicinal plants including Chinese herbs. Among these iridoid glycosides, AU was systematically studied for its potent liver-protective activities using experimental systems of hepatic damage. AU showed high liver-protective activity against carbon tetrachloride-induced hepatic damage in mice. Also AU showed significant protective activity against alpha-amanitin-induced hepatic damage in mice, and it prevented a depression of liver RNA biosynthesis caused by alpha-amanitin administration. Potent antidotal effects on mushroom poisoning in beagle dogs ingested with aqueous extract of Amanita virosa was observed; beagle dogs completely survived, even when AU administration was withheld for half an hour after mushroom poisoning. In addition, AU was found to suppress hepatitis B viral DNA replication in vitro. Conversion of AU to its aglycone form appeared to be a prerequisite step for an exhibition of such antiviral activity.
Int Immunopharmacol. 2015 Feb;24(2):408-15.
Aucubin prevents interleukin-1 beta induced inflammation and cartilage matrix degradation via inhibition of NF-κB signaling pathway in rat articular chondrocytes.[Pubmed:
25576403]
Proinflammatory cytokine interleukin-1β (IL-1β) plays a crucial role in the pathogenesis of Osteoarthritis (OA) by stimulating several mediators contributed to cartilage degradation.
Aucubin, a natural compound derived from plants which has been shown to possess diverse biological activities including anti-inflammatory property, may benefit the IL-1β stimulated chondrocytes.
METHODS AND RESULTS:
The present study was aimed to investigate the effects of Aucubin on IL-1β stimulated rat chondrocytes. Rat chondrocytes were cultured and pretreated with Aucubin (1, 10, 20, 50μM), and then stimulated with or without IL-1β (10ng/ml). Gene and protein expression of MMP-3, MMP-9, MMP-13, cyclooxygenase-2 (COX-2), inducible nitric oxide synthase (iNOS) was determined by real-time PCR and Western blotting respectively. Nitric oxide (NO) production was quantified by Griess reagent. Phosphorylation and nuclear translocation of p65 were detected by western blotting and immunofluorescence, respectively. We found that Aucubin significantly reversed the elevated gene and protein expression of MMP-3, MMP-9, MMP-13, iNOS, COX-2 and the production of NO induced by IL-1β challenge in rat chondrocytes. Furthermore, Aucubin was able to suppress the IL-1β-mediated phosphorylation and nuclear translocation of p65, indicating Aucubin may possibly act via the NF-κB signaling pathway.
CONCLUSIONS:
The present study proposes that Aucubin may be a potential therapeutic choice in the treatment of OA due to its anti-inflammatory and chondroprotective features.
Inflammation . 2017 Dec;40(6):2062-2073.
Aucubin Alleviates Bleomycin-Induced Pulmonary Fibrosis in a Mouse Model[Pubmed:
28785877]
Abstract
Pulmonary fibrosis is a life-threatening disease characterized by progressive dyspnea and worsening of pulmonary function. No effective therapeutic strategy for pulmonary fibrosis has been established. Aucubin is a natural constituent with a monoterpene cyclic ring system. The potency of Aucubin in protecting cellular components against inflammation, oxidative stress, and proliferation effects is well documented. In this study, we investigated the protective effect of Aucubin against pulmonary fibrosis in mice. A mouse model of pulmonary fibrosis was established by intratracheal injection of bleomycin (BLM), and Aucubin was administered for 21 days after BLM injection. We found that Aucubin decreased the breathing frequency and increased the lung dynamic compliance of BLM-stimulated mice detected by Buxco pulmonary function testing system. Histological examination showed that Aucubin alleviated BLM-induced lung parenchymal fibrotic changes. Aucubin also reduced the intrapulmonary collagen disposition and inflammatory injury induced by BLM. In addition, Aucubin reduced the expression of pro-fibrotic protein transforming growth factor (TGF)-β1 and α-smooth muscle actin (α-SMA) of pulmonary fibrosis mice induced by BLM. Furthermore, the effect of Aucubin on the proliferation and differentiation of fibroblast was investigated in vitro. Aucubin inhibited the mRNA and protein expression of Ki67 and proliferating cell nuclear antigen (PCNA) induced by TGF-β1 and reduced the cell proliferation in a murine fibroblast cell NIH3T3. Aucubin also reduced the collagen syntheses and α-SMA expression induced by TGF-β1 in fibroblast. Our results indicate that Aucubin inhibits inflammation, fibroblast proliferation, and differentiation, exerting protective effects against BLM-induced pulmonary fibrosis in a mouse model. This study provides an evidence that Aucubin may be a novel drug for pulmonary fibrosis.
Planta Med. 1994 Dec;60(6):512-5.
In vitro antispasmodic activity of peracetylated penstemonoside, aucubin and catalpol.[Pubmed:
7809202]
In vitro, the peracetates of penstemonoside, Aucubin and catalpol, iridoids isolated from Parentucellia latifolia, antagonize the uterine muscular contractions induced by acetylcholine and calcium, in a similar way to papaverine.
METHODS AND RESULTS:
The antagonism is non-competitive against acetylcholine (pD2' values: 5.60 for the peracetates of penstemonoside, 5.74 for Aucubin, 5.59 for catalpol, and 5.32 for papaverine) and competitive against calcium (pA2 values: 6.60 for the peracetates of penstemonoside, 6.34 for Aucubin, 6.48 for catalpol, and 6.23 for papaverine).
The two components, phasic and tonic, of the response of the vas deferens to potassium are reduced by the three iridoids.
CONCLUSIONS:
The reduction is similar for both phases.
The antispasmodic activity of the three iridoids, similar to papaverine, is related to an inhibiting effect of extracellular calcium, intracellular or both.
Biol Pharm Bull. 2005 Jul;28(7):1244-8.
Protective effects of aucubin isolated from Eucommia ulmoides against UVB-induced oxidative stress in human skin fibroblasts.[Pubmed:
15997107]
Ultraviolet-B (UVB) irradiation has been demonstrated to produce reactive oxygen species (ROS) in the cells and skin, which induces the synthesis of matrix metalloproteinases (MMPs), causing skin photoaging.
METHODS AND RESULTS:
Using the human skin fibroblast HS68 cell line in the present study, we investigated the photoprotective effects of Aucubin from Eucommia ulmoides.
Pretreatment with Aucubin significantly inhibited the production of MMP-1 by 57% when compared to the UVB-irradiated cells. Additionally, the senescence-associated beta-galactosidase (SA beta-gal) activity was markedly decreased in the presence of Aucubin, which indicates it as an antiphoto-induced aging compound. As the effect of Aucubin was determined against ROS, the inhibited ROS formation and malondialdehyde (MDA) levels, and the increased cell viability and glutathione (GSH) level were observed with Aucubin under UVB irradiation.
METHODS AND RESULTS:
Based upon these results, it was suggested that Aucubin might play an important role in the cellular defense mechanism against UV radiation-induced photoaging. An understanding of the antioxidant properties of Aucubin could, in part, act to elucidate its protective mechanism on the human skin photoaging.
Eur J Pharmacol. 2008 Mar 17;582(1-3):162-7.
Antioxidant and pancreas-protective effect of aucubin on rats with streptozotocin-induced diabetes.[Pubmed:
18230397]
Oxidative stress has been suggested as a contributory factor in development and complication of diabetes.
The aim of the present study was to determine the protective effect of Aucubin on lipid peroxidation and activities of antioxidant defense systems and to conduct immunohistochemical evaluation of pancreas in streptozotocin-induced diabetic rats.
METHODS AND RESULTS:
Lipid peroxidation was determined by assessing the concentration of malondialdehyde and activities of antioxidant enzymes - catalase, glutathione peroxidase and superoxide dismutase in liver and kidneys of rats were determined. Changes of blood glucose and immunohistochemical evaluation on pancreas were also investigated as part of the pathology of diabetes. In our study, Aucubin treatment lowered blood glucose. Diabetic rats exhibited an increase in the level of lipid peroxidation and decrease in activities of antioxidant enzymes in liver and kidneys as compared to control rats. Administration of Aucubin to diabetic rats for 15 days significantly reversed damage associated with diabetes. In addition, diabetic rats showed an obvious decrease in insulin immunoreactivity and the number of beta cells in pancreas, but the pancreas of Aucubin-treated rats were improved and the number of immunoreactive beta cells were significantly increased.
CONCLUSIONS:
These results indicated that Aucubin may have value as a safe preventive or therapeutic agent against diabetes mellitus.
Biol Pharm Bull. 1998 Dec;21(12):1306-10.
The promoting effects of geniposidic acid and aucubin in Eucommia ulmoides Oliver leaves on collagen synthesis.[Pubmed:
9881644]
We have reported that collagen synthesis was stimulated by the administration of a hot water extract from the leaves of Eucommia ulmoides OLIVER, Eucommiaceae (Du-Zhong leaves) in false aged model rats.
METHODS AND RESULTS:
In this paper, we set out to examine the compounds in Du-Zhong leaves that stimulated collagen synthesis in false aged model rats. In experiment 1, a methanol extract of Du-Zhong leaves also stimulated collagen synthesis in aged model rats. An acetone fraction was derived from the methanol extract by silica gel chromatography in experiment 2. The acetone fraction mainly contained iridoides mono-glycosides such as geniposidic acid and Aucubin. The administration of geniposidic acid or Aucubin stimulated collagen synthesis in aged model rats in experiments 3 and 4 (significance (p<0.05)).
CONCLUSIONS:
The reported pharmacological effects of Du-Zhong leaves, including healing organs and strengthening bone and muscle, are closely related to collagen metabolism. It appears that geniposidic acid and Aucubin are the actual compounds in Du-Zhong which caused the effect in our experiments.