Torilin
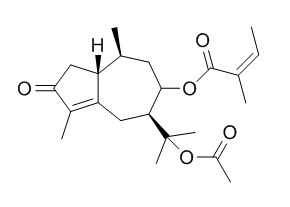
Torilin is an inhibitor of testosterone 5 alpha-reductase, it (IC50 = 31.7 +/- 4.23 microM) shows a stronger inhibition of 5 alpha-reductase than alpha-linolenic acid (IC50 = 160.3 +/- 24.62 microM) but is weaker than finasteride (IC50 = 0.38 +/- 0.06 microM). Torilin has immunomodulatory, hepatoprotective, and anti-inflammatory properties, it inhibits inflammation by limiting TAK1-mediated MAP kinase and NF-κB activation, it can attenuate arthritis severity, modify leukocyte activations in dLNs or joints, and restore serum and splenocyte cytokine imbalances. Torilin inhibits melanin production in alpha-melanocyte stimulating hormone-activated B16 melanoma cells, with an IC(50) value of 25 microM. Torilin shows excellent antimicrobial activity against Bacillus subtilis ATCC 6633 spores and vegetative cells. Torilin has a potent anti-angiogenic activity both in vivo and in vitro, and it may have a strong activity to suppress tumorigenesis by inhibition of tumor invasion, it reverses multidrug-resistance in cancer cells, it can potentiate the cytotoxicities of adriamycin, vinblastine, taxol and colchicine against multidrug-resistant KB-V1 and MCF7/ADR cells.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Int J Med Sci.2021, 18(10):2155-2161.
Food Sci Nutr.2023, 11(9):5532-5542.
South African J of Plant&Soil2018, 29-32
J Ethnopharmacol.2017, 206:73-77
Oncotarget.2015, 6(31):30831-49
Eur Rev Med Pharmacol Sci.2020, 24(9):5127-5139.
National Academy Science Letters2023, s40009.
Korean J. Medicinal Crop Sci.2021, 29(6):425-433
Phytother Res.2022, 10.1002:ptr.7626.
Phytomedicine.2019, 58:152893
Related and Featured Products
Planta Med. 2009 Nov;75(14):1505-8.
Torilin from Torilis japonica inhibits melanin production in alpha-melanocyte stimulating hormone-activated B16 melanoma cells.[Pubmed:
19533579 ]
Epidermal melanocytes synthesize melanin pigments and transfer them to keratinocytes, which is responsible for skin pigmentation. However, abnormal accumulation of melanin pigments causes hyperpigmentation disorders, which are substantially improved with treatment of tyrosinase inhibitor. In our ongoing study, Torilis japonica DC. (Umbelliferae) was found to inhibit melanin production.
A goal of this study is to elucidate the hypopigmenting principle of T. japonica.
METHODS AND RESULTS:
A sesquiterpene structure of Torilin was isolated from the plant extracts via bioassay-guided phytochemical analysis. Torilin dose-dependently inhibited melanin production, with an IC(50) value of 25 microM, in alpha-melanocyte stimulating hormone (alpha-MSH)-activated B16 melanoma cells. Arbutin, a positive control of skin whitener, also inhibited alpha-MSH-induced melanin production with an IC(50) value of 170 microM. As to the mode of action, Torilin downregulated alpha-MSH-induced protein levels of tyrosinase without directly inhibiting catalytic activity of the enzyme.
CONCLUSIONS:
Taken together, this study shows that Torilin contributes to the hypopigmenting principle of T. japonica, and suggests its pharmacological potential in melanin-associated hyperpigmentation disorders.
J Food Sci. 2008 Mar;73(2):M37-46.
Antimicrobial activity of torilin isolated from Torilis japonica fruit against Bacillus subtilis.[Pubmed:
18298734 ]
Torilis japonica fruit has been used in therapeutic antimicrobial treatments in Korea and China since ancient times, but there is still little information on the mechanism underlying this activity.
METHODS AND RESULTS:
We found that the ethanol extract of T. japonica fruit showed excellent antimicrobial activity against Bacillus subtilis ATCC 6633 spores and vegetative cells. The crude ethanol extract (75%) reduced the spore concentration by 3 log cycles and the vegetative cell concentration to lower than the detection level (reduction in spore concentration by more than 6 orders of magnitude) at a concentration of 1% (w/v). The ethanol extract of T. japonica fruit was fractionated into n-hexane (H) and a water layer. The active antimicrobial compound was isolated and purified from the hexane layer, and identified as Torilin (5-[1-(acetyloxy)-1-methylethyl]-3,8-dimethyl-2-oxo-1,2,4,5,6,7,8,8a-octa-hydroazulen-6-yl(2E)-2-methylbut-2-enoate; C(22)H(32)O(5)). Torilin immediately reduced vegetative cells counts by 5 to 6 orders of magnitude, and reduced spores counts by 1 order of magnitude. The integrity of structures such as the inner, middle, and outer layers of the coat and the cortex, protoplast membrane, and core are vital to spores.
CONCLUSIONS:
Torilin functions as a surfactant with hydrophilic and hydrophobic properties related to denaturalization of various proteins.
The distortion of coat proteins due to direct binding polar groups of spore coats with hydrophilic groups of Torilin may be responsible for the observed rapid inactivation of bacterial spores.
Planta Med. 2002 Aug;68(8):748-9.
Sesquiterpenes with hepatoprotective activity from Cnidium monnieri on tacrine-induced cytotoxicity in Hep G2 cells.[Pubmed:
12221602 ]
METHODS AND RESULTS:
Bioassay-guided fractionation of the EtOH extract of Cnidium monnieri (Apiaceae) furnished two hepatoprotective sesquiterpenes, Torilin (1) and torilolone (2), together with a new derivative, 1-hydroxyTorilin (3).
CONCLUSIONS:
Compounds 1 and 2 showed hepatoprotective effects on tacrine-induced cytotoxicity in human liver-derived Hep G2 cells. The EC50 values of compounds 1 and 2 were 20.6 +/- 1.86 (P < 0.01) and 3.6 +/- 0.1 (P < 0.01) microM, respectively. Silybin as a positive control showed an EC50 value of 69.0 +/- 3.4 microM.
Int Immunopharmacol. 2013 Jun;16(2):232-42.
Torilin ameliorates type II collagen-induced arthritis in mouse model of rheumatoid arthritis.[Pubmed:
23623942 ]
Advancements in rheumatoid-arthritis-(RA) therapies have shown considerable progresses in the comprehension of disease. However, the development of new potential agents with relative safety and efficacy continues and natural compounds have been considered as alternatives to identify new entities.
METHODS AND RESULTS:
Since previous in-vivo data and our in-vitro findings showed that Torilin has a strong anti-inflammatory property, we further investigated its effect against collagen-induced-arthritis-(CIA) in mice. CIA-induced DBA/1J mice were treated with Torilin or methotrexate (MTX) for 5-weeks. Arthritis severity was evaluated by arthritic score and joint histopathology. Draining lymph node (dLN), joint and peripheral-blood mononuclear-cell (PBMC) counts, and activation/localization of T-/B-lymphocytes, dendritic cells (DCs) and neutrophils were examined by FACS analysis. Serum anti-type-II-collagen-(CII) antibody levels and cultured-splenocyte and serum cytokines were also evaluated. Torilin markedly reduced CIA-induced arthritic score, histopathology and leukocyte counts. Besides, Torilin suppressed CIA-activated T-cells including CD3+, CD3+/CD69+, CD8+, CD4+ and CD4+/CD25+ in dLNs or joints. It also modified CD19+ or CD20+/CD23+ (B-cells), MHCII+/CD11c+ (DCs) and Gr-1+/CD11b+ (neutrophil) subpopulations. It further depressed total anti-CII-IgG, anti-CII-IgG1 and anti-CII-IgG2a antibody productions. Moreover, while IFN-γ and IL-10 were not affected, Torilin suppressed CIA-induced serum TNF-α, IL-1β and IL-6 levels. Interestingly, Torilin also blocked IFN-γ, IL-17 and IL-6 cytokines while it did not affect IL-10 but enhanced IL-4 in splenocytes.
CONCLUSIONS:
These results show that Torilin attenuated arthritis severity, modified leukocyte activations in dLNs or joints, and restored serum and splenocyte cytokine imbalances. Torilin may have immunomodulatory and anti-inflammatory properties with the capacity to ameliorate the inflammatory response in CIA-mice.
Oncol Rep. 2001 Mar-Apr;8(2):359-64.
Anti-invasive activity of torilin, a sesquiterpene compound isolated from Torilis japonica.[Pubmed:
11182056]
Torilin is a sesquiterpene compound purified from Torilis japonica (Umbelliferae). We have previously reported that Torilin has a potent anti-angiogenic activity both in vivo and in vitro.
METHODS AND RESULTS:
In the present study, we investigated the anti-invasive activity of Torilin, and interestingly found that Torilin completely blocked intravasation of HT1080 human fibrosarcoma cells inoculated on the chorioallantoic membrane (CAM) of chick embryo. In addition, Torilin decreased the attachment of HT1080 cells to confluent human umbilical vein endothelial cells (HUVECs) at non-toxic concentration. In in vitro transwell invasion model, 25 microM Torilin also significantly inhibited HT1080 cell invasion in a time-dependent manner. Activity and expression of matrix metalloproteinase-9 (MMP-9) that is very important in tumor invasion and metastasis were also decreased by Torilin treatment, indicating that the inhibitory effect of Torilin on invasion of HT1080 cells may result from decreasing activity and expression of MMP-9.
CONCLUSIONS:
Therefore, it is possible that Torilin may decrease metastatic potential of tumor cells through inhibiting their attachment to endothelial cells and intravasation to blood vessels. Taken together, Torilin may have a strong activity to suppress tumorigenesis by inhibition of tumor invasion.
Mediators Inflamm. 2017;2017:7250968.
Torilin Inhibits Inflammation by Limiting TAK1-Mediated MAP Kinase and NF-κB Activation.[Pubmed:
28316375 ]
Torilin, a sesquiterpene isolated from the fruits of Torilis japonica, has shown antimicrobial, anticancer, and anti-inflammatory properties. However, data on the mechanism of Torilin action against inflammation is limited. This study aimed at determining the anti-inflammatory property of Torilin in LPS-induced inflammation using in vitro model of inflammation.
METHODS AND RESULTS:
We examined Torilin's effect on expression levels of inflammatory mediators and cytokines in LPS-stimulated RAW 264.7 macrophages. The involvement of NF-kB and AP-1, MAP kinases, and adaptor proteins were assessed. Torilin strongly inhibited LPS-induced NO release, iNOS, PGE2, COX-2, NF-α, IL-1β, IL-6, and GM-CSF gene and protein expressions. In addition, MAPKs were also suppressed by Torilin pretreatment. Involvement of ERK1/2, P38MAPK, and JNK1/2 was further confirmed by PD98059, SB203580, and SP600125 mediated suppression of iNOS and COX-2 proteins. Furthermore, Torilin attenuated NF-kB and AP-1 translocation, DNA binding, and reporter gene transcription. Interestingly, Torilin inhibited TAK1 kinase activation with the subsequent suppression of MAPK-mediated JNK, p38, ERK1/2, and AP-1 (ATF-2 and c-jun) activation and IKK-mediated I-κBα degradation, p65/p50 activation, and translocation.
CONCLUSIONS:
Together, the results revealed the suppression of NF-κB and AP-1 regulated inflammatory mediator and cytokine expressions, suggesting the test compound's potential as a candidate anti-inflammatory agent.
Planta Med. 2003 May;69(5):459-61.
Torilin from Torilis japonica, as a new inhibitor of testosterone 5 alpha-reductase.[Pubmed:
12802730 ]
The methanolic extract of the fruits of Torilis japonica showed a potent inhibition against 5 alpha-reductase activity in vitro.
METHODS AND RESULTS:
Bioassay-guided fractionation of the methanol extract of the fruits followed by repeated silica gel chromatography led to the isolation of an active principle and its structure was identified as Torilin on the basis of spectroscopic data. Torilin (IC50 = 31.7 +/- 4.23 microM) showed a stronger inhibition of 5 alpha-reductase than alpha-linolenic acid (IC50 = 160.3 +/- 24.62 microM) but was weaker than finasteride. (IC50 = 0.38 +/- 0.06 microM). Simple guaiane-type compounds, such as (-)-guaiol and guaiazulene showed weak inhibitory effects on the 5 alpha-reductase activity with IC50 values of f 81.6 microM and 100.8 microM, respectively, while azulene was not active.
CONCLUSIONS:
These results suggest that both degrees of unsaturation and the side-chain in the guaiane skeleton are important for the manifestation of 5 alpha-reductase inhibition.
Planta Med. 1998 May;64(4):332-4.
Torilin, a sesquiterpene from Torilis japonica, reverses multidrug-resistance in cancer cells.[Pubmed:
9619115]
METHODS AND RESULTS:
A sesquiterpene compound reversing multidrug-resistance in cancer cells was isolated from the fruits of Torilis japonica and the structure was identified as Torilin. Torilin potentiated the cytotoxicities of adriamycin, vinblastine, taxol and colchicine against multidrug-resistant KB-V1 and MCF7/ADR cells.



