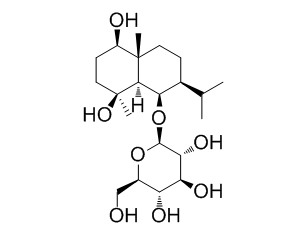
Ophiopogonoside A
Reference standards.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Environ Toxicol.2023, 23929.
Korean j.of Pharm.2017, 70-76
Journal of Medicinal Food2023, Vol.26(10).
Sci Rep.2019, 9(1):18080
Evid Based Complement Alternat Med.2017, 2017:7383104
Int J Mol Sci.2023, 24(20):15320.
Int. J. Mol. Sci.2022, 23(8), 4130.
Toxicol In Vitro.2023, 93:105667.
PLoS One.2018, 13(11):e0208055
Antiviral Res.2013, 98(3):386-93
Related and Featured Products
J Asian Nat Prod Res. 2012;14(5):491-5.
A new eudesmane sesquiterpene glycosides from Liriope muscari.[Pubmed:
22423627]
METHODS AND RESULTS:
A new eudesmane sesquiterpene glycoside, ophiopogonoside B (1), along with five known compounds, Ophiopogonoside A (2), ruscogenin-1-O-[β-D-glucopyranosyl (1 → 2)]-[β-D-xylopyranosyl (1 → 3)]-β-D-fucopyranoside (3), palmitic acid (4), palmitic acid glyceride (5), and β-sitosterol-D-glucopyranoside (6),was isolated from the tuberous roots of Liriope muscari (Decn.) Bailey (Liliaceae). Their structures were confirmed by 1D and 2D NMR spectroscopy.
CONCLUSIONS:
Among them, compounds 1, 2, 4, and 5 were the first reported from the genus Liriope. Ophiopogonoside B (1) showed moderate inhibitory activity to glycogen phosphorylase a.
J Nat Prod. 2004 Oct;67(10):1761-3.
cis-Eudesmane sesquiterpene glycosides from Liriope muscari and Ophiopogon japonicus.[Pubmed:
15497959]
METHODS AND RESULTS:
Two new cis-eudesmane sesquiterpene glycosides, liriopeoside A (1) and Ophiopogonoside A (2), were extracted and purified from tubers of Liriope muscari and Ophiopogon japonicus, respectively, along with three known compounds. Their structures were elucidated as 1beta,6beta-dihydroxy-cis-eudesm-3-ene-6-O-beta-D-glucopyranoside (1) and 1beta,4beta,6beta-trihydroxy-cis-eudesmane-6-O-beta-D-glucopyranoside (2) by spectral data analysis. The structure and the relative configuration of compound 1 were confirmed by X-ray crystallographic analysis.
CONCLUSIONS:
This is the first time that cis-eudesmane-type sesquiterpenes have been reported from the genera Ophiopogon and Liriope.