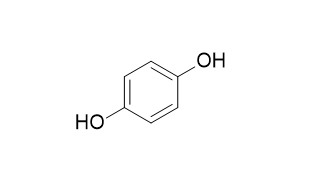
Hydroquinone
Hydroquinone is a nonvolatile chemical used in the photographic, rubber, chemical, and cosmetic industries. Hydroquinone is one of the most effective inhibitors of melanogenesis in vitro and in vivo, and is widely used for the treatment of melanosis and other hyperpigmentary disorders, the depigmenting activity of it may partly be related to the ability of the compound to act as an alternate substrate of tyrosinase, thereby competing for tyrosine oxidation in active melanocytes.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Appl. Sci.2022, 12(4), 2032.
Drug Dev Res.2022, 83(7):1673-1682.
Antiviral Res.2013, 98(3):386-93
Universite de Bordeaux2017, 2017BORD0867
Biochem Biophys Res Commun.2021, 534:802-807.
Molecules.2020, 25(18):4283.
Molecules.2023, 28(5):2376.
Journal of Mushroom2024, 22(4):192-198
Vietnam Journal of Science2022, 64(2), 69-75.
Food Res Int.2020, 133:109130.
Related and Featured Products
Biochimica Et Biophysica Acta General Subjects, 1991,1073(1): 85-90.
Mechanism of inhibition of melanogenesis by hydroquinone.[Reference:
WebLink]
Hydroquinone (HQ) is one of the most effective inhibitors of melanogenesis in vitro and in vivo, and is widely used for the treatment of melanosis and other hyperpigmentary disorders.
METHODS AND RESULTS:
In an attempt to get some insight into the molecular mechanism of the depigmenting action, which is still very poorly understood, we have investigated the effect of HQ on the tyrosinase catalysed conversion of tyrosine to melanin. Incubation of 0.5 mM tyrosine with 0.07 U/ml tyrosinase in phosphate buffer at pH 6.8 in the presence of 0.5 mM HQ led to no detectable melanin formation, due to the preferential oxidation of HQ with respect to tyrosine (HPLC evidence). Kinetic investigations showed that HQ is a poorer substrate of tyrosinase than tyrosine; yet, it may be effectively oxidised in the presence of tyrosine owing to the generation of catalytic amounts of dopa acting as cofactor of tyrosinase. Product analysis of HQ oxidation with tyrosinase in the presence of dopa showed the predominant formation in the early stages of hydroxybenzoquinone (HBQ), arising from enzymic hydroxylation and subsequent oxidation of HQ, along with lower amounts of benzoquinone (BQ).
CONCLUSIONS:
These results suggest that the depigmenting activity of HQ may partly be related to the ability of the compound to act as an alternate substrate of tyrosinase, thereby competing for tyrosine oxidation in active melanocytes.
J Toxicol Environ Health, 1996, 47(1):31-46.
Human exposure to naturally occurring hydroquinone.[Pubmed:
8568910]
Hydroquinone (HQ) is a nonvolatile chemical used in the photographic, rubber, chemical, and cosmetic industries. HQ is also known to occur in nature as the beta-D-glucopyranoside conjugate (arbutin), and free HQ is a known component of cigarette smoke. Low concentrations of HQ have been detected in the urine and plasma of humans with no occupational or other known exposure to HQ.
METHODS AND RESULTS:
The studies reported here investigate dietary and other potential sources of HQ and their contribution to HQ concentrations in the plasma and urine of human volunteers. Analysis of possible food sources of HQ by GC indicated significant amounts of arbutin in wheat products (1-10 ppm), pears (4-15 ppm), and coffee and tea (0.1 ppm). Free HQ was found in coffee (0.2 ppm), red wine (0.5 ppm), wheat cereals (0.2-0.4 ppm), and broccoli (0.1 ppm). After consuming a meal including arbutin- and HQ-containing foods, volunteers showed significant increases in plasma and urinary levels of HQ and its conjugated metabolites (total HQ). Mean plasma concentrations of total HQ peaked at 5 times background levels at 2 h after the completion of the meal, and mean urinary excretion rates of total HQ peaked at 12 times background at 2-3 h after the meal. Immediately after smoking four cigarettes in approximately 30 min, mean plasma concentrations of total HQ were maximally 1.5 times background levels; mean urinary excretion rates of total HQ peaked at 2.5 times background at 1-3 h after smoking.
CONCLUSIONS:
These data indicate that considerable human exposure to HQ can result from plant-derived dietary sources and, to a lesser extent, from cigarette smoke.