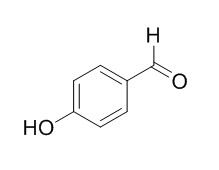
4-Hydroxybenzaldehyde
4-Hydroxybenzaldehyde shows an inhibitory effect on the GABA transaminase to contribute to an antiepileptic and anticonvulsive activity, and its inhibitory activity was higher than that of valproic acid, a known anticonvulsant.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Antioxidants (Basel).2020, 9(7):581.
Chemistry of Natural Compounds2018, 54(3):572-576
Molecules2022, 27(11):3606.
Foods.2022, 11(12):1773.
J Int Med Res.2021, 49(7):3000605211032849.
Fitoterapia.2024, 175:105958.
J of Archaeological Science:Reports2024, 53:104298
Biochem Biophys Res Commun.2018, 495(1):1271-1277
Food Chem X.2024, 24:101909.
Journal of Food and Drug Analysis2023, 31(3), 9.
Related and Featured Products
Malays J Med Sci . 2017 Mar;24(2):94-99.
The Effect of 4-hydroxybenzaldehyde on the γ-aminobutyric Acid Type A Receptor[Pubmed:
28894409]
Abstract
The α1β2γ2 subtype of GABAA receptors is the most commonly found GABAA receptor subtype in the mammalian cortex and hippocampus. It is expressed heterologously in the Xenopus laevis oocyte as a α1β2γ2S/L subtype for application as an in vitro model for the screening of compounds that modulate receptor activities. In fact, 4-Hydroxybenzaldehyde (4-HB) has been identified as one of the major components in Dendrocalamus asper bamboo shoots in our previous study, and the current study showed that at 101.7 μM, 4-HB significantly reduced the GABA-induced chloride current of GABAA receptors expressed on Xenopus oocytes, indicating a possible GABAergic antagonistic effect at high concentrations.
Keywords: GABAergic agents; Xenopus oocytes; bamboo shoot; electrophysiology; α1β2γ2 GABAA receptor.
Bioorg Med Chem Lett. 2006 Feb;16(3):592-5. Epub 2005 Nov 14.
Inhibition of GABA shunt enzymes' activity by 4-hydroxybenzaldehyde derivatives.[Pubmed:
16290145]
4-Hydroxybenzaldehyde (HBA) derivatives were examined as inhibitors for GABA transaminase (GABA-T) and succinic semialdehyde dehydrogenase (SSADH). Investigation of structure-activity relation revealed that a carbonyl group or an amino group as well as a hydroxy group at the para position of the benzene ring are important for both enzymes' inhibition. HBA was shown to give competitive inhibition of GABA-T with respect to alpha-ketoglutarate and competitive inhibition of SSADH. 4-Hydroxybenzylamine (HBM) also showed the competitive inhibition on GABA-T with respect to GABA.
Biochemistry. 2015 Feb 10;54(5):1219-32.
Functional and structural characterization of an unusual cofactor-independent oxygenase.[Pubmed:
25565350]
The vast majority of characterized oxygenases use bound cofactors to activate molecular oxygen to carry out oxidation chemistry. Here, we show that an enzyme of unknown activity, RhCC from Rhodococcus jostii RHA1, functions as an oxygenase, using 4-hydroxyphenylenolpyruvate as a substrate. This unique and complex reaction yields 3-hydroxy-3-(4-hydroxyphenyl)-pyruvate, 4-Hydroxybenzaldehyde, and oxalic acid as major products. Incubations with H2(18)O, (18)O2, and a substrate analogue suggest that this enzymatic oxygenation reaction likely involves a peroxide anion intermediate. Analysis of sequence similarity and the crystal structure of RhCC (solved at 1.78 Å resolution) reveal that this enzyme belongs to the tautomerase superfamily. Members of this superfamily typically catalyze tautomerization, dehalogenation, or decarboxylation reactions rather than oxygenation reactions. The structure shows the absence of cofactors, establishing RhCC as a rare example of a redox-metal- and coenzyme-free oxygenase. This sets the stage to study the mechanistic details of cofactor-independent oxygen activation in the unusual context of the tautomerase superfamily.