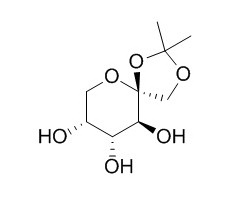
1,2-O-Isopropylidene-beta-D-fructopyranose
Standard reference
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Int Immunopharmacol.2024, 143(Pt 2):113486.
Auburn University2015, 1-58
Reprod Toxicol.2020, 96:1-10.
J Ethnopharmacol.2017, 198:91-97
Oncotarget.2016, 8(51):88386-88400
Molecules.2022, 27(22):7997.
J AOAC Int.2023, 106(1):56-64.
Genes (Basel).2021, 12(7):1024.
Aquaculture2019, 510:392-399
Drug Des Devel Ther.2020, 14:969-976.
Related and Featured Products
J Org Chem. 2014 Oct 17;79(20):9444-50.
Synthesis of a poly-hydroxypyrolidine-based inhibitor of Mycobacterium tuberculosis GlgE.[Pubmed:
25137149]
Long treatment times, poor drug compliance, and natural selection during treatment of Mycobacterium tuberculosis (Mtb) have given rise to extensively drug-resistant tuberculosis (XDR-TB). As a result, there is a need to identify new antituberculosis drug targets.
METHODS AND RESULTS:
Mtb GlgE is a maltosyl transferase involved in α-glucan biosynthesis. Mutation of GlgE in Mtb increases the concentration of maltose-1-phosphate (M1P), one substrate for GlgE, causing rapid cell death. We have designed 2,5-dideoxy-3-O-α-d-glucopyranosyl-2,5-imino-d-mannitol (9) to act as an inhibitor of GlgE. Compound 9 was synthesized using a convergent synthesis by coupling thioglycosyl donor 14 and 5-azido-3-O-benzyl-5-deoxy-1,2-O-Isopropylidene-beta-D-fructopyranose (23) to form disaccharide 24. A reduction and intramolecular reductive amination transformed the intermediate disaccharide 24 to the desired pyrolidine 9. Compound 9 inhibited both Mtb GlgE and a variant of Streptomyces coelicolor (Sco) GlgEI with Ki = 237 ± 27 μM and Ki = 102 ± 7.52 μM, respectively.
CONCLUSIONS:
The results confirm that a Sco GlgE-V279S variant can be used as a model for Mtb GlgE. In conclusion, we designed a lead transition state inhibitor of GlgE, which will be instrumental in further elucidation of the enzymatic mechanism of Mtb GlgE.