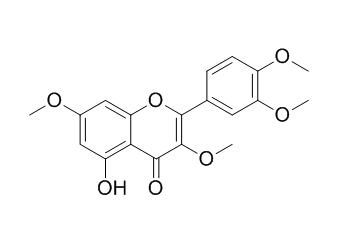
Retusin
Retusin behaves as pre- and postemergent herbicides. Retusin is a weak inhibitor of ATP synthesis; it exhibits potent free-radical-scavenging capacities as well as efficient inhibition of cellular melanogenesis.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Int J Mol Sci.2022, 23(5):2796.
Biomed Pharmacother.2022, 145:112410.
Antioxidants.2022, 11(3):491.
Evid Based Complement Alternat Med.2020, 2020:2584783.
Journal of Ginseng Research2022, j.jgr.2022.09.005.
Int Immunopharmacol.2023, 125:111175.
Am J Chin Med.2022, 1-20.
Hum. Ecol. Res.2025, 63(2):165-174
Molecules.2020, 25(21):5091.
Kor. J. Pharmacogn.2016, 47(1):62-72
Related and Featured Products
J Agric Food Chem. 2015 Sep 23;63(37):8106-15.
Flavonoids Affect the Light Reaction of Photosynthesis in Vitro and in Vivo as Well as the Growth of Plants.[Pubmed:
26322527 ]
Flavonoids Retusin (5-hydroxy-3,7,3',4'-tetramethoxyflavone) (1) and pachypodol (5,4'-dihydroxy-3,7,3'-trimethoxyflavone) (2) were isolated from Croton ciliatoglanduliferus Ort.
METHODS AND RESULTS:
Pachypodol acts as a Hill reaction inhibitor with its target on the water splitting enzyme located in PSII. In the search for new herbicides from natural compounds, flavonoids 1 and 2 and flavonoid analogues quercetin (3), apigenin (4), genistein (5), and eupatorin (6) were assessed for their effect in vitro on the photosynthetic electron transport chain and in vivo on the germination and growth of the plants Physalis ixocarpa, Trifolium alexandrinum and Lolium perenne. Flavonoid 3 was the most active inhibitor of the photosynthetic uncoupled electron flow (I50 = 114 μM) with a lower log P value (1.37).
CONCLUSIONS:
Results in vivo suggest that 1, 2, 3, and 5 behave as pre- and postemergent herbicides, with 3 and 5 being more active.
Chem Biodivers. 2015 Jun;12(6):963-79.
Free-Radical-Scavenging, Antityrosinase, and Cellular Melanogenesis Inhibitory Activities of Synthetic Isoflavones.[Pubmed:
26080742]
In this study, we examined the potential of synthetic isoflavones for application in cosmeceuticals.
METHODS AND RESULTS:
Twenty-five isoflavones were synthesized and their capacities of free-radical-scavenging and mushroom tyrosinase inhibition, as well as their impact on cell viability of B16F10 murine melanoma cells and HaCaT human keratinocytes were evaluated. Isoflavones that showed significant mushroom tyrosinase inhibitory activities were further studied on reduction of cellular melanin formation and antityrosinase activities in B16F10 melanocytes in vitro. Among the isoflavones tested, 6-hydroxydaidzein (2) was the strongest scavenger of both ABTS(.+) and DPPH(.) radicals with SC50 values of 11.3 ± 0.3 and 9.4 ± 0.1 μM, respectively. Texasin (20) exhibited the most potent inhibition of mushroom tyrosinase (IC50 14.9 ± 4.5 μM), whereas Retusin (17) showed the most efficient inhibition both of cellular melanin formation and antityrosinase activity in B16F10 melanocytes, respectively.
CONCLUSIONS:
In summary, both Retusin (17) and texasin (20) exhibited potent free-radical-scavenging capacities as well as efficient inhibition of cellular melanogenesis, suggesting that they are valuable hit compounds with potential for advanced cosmeceutical development.
J Agric Food Chem. 2006 Feb 22;54(4):1217-21.
Pachypodol from Croton ciliatoglanduliferus Ort. as water-splitting enzyme inhibitor on thylakoids.[Pubmed:
16478239]
METHODS AND RESULTS:
A bioactivity-guided chemical study of aerial parts of Croton ciliatoglanduliferus Ort. led to the isolation for the first time of the flavonoids Retusin (5-hydroxy-3,7,3',4'-tetramethoxyflavone) (1) and pachypodol (5,4'-dihydroxy-3,7,3'-trimethoxyflavone) (2) from the n-hexane extract.
Compounds 1 and 2 were separated by preparative thin-layer chromatography. Compound 2 was the most active compound on ATP synthesis inhibition. The I50 value was 51 microM. Pachypodol behaves as a Hill reaction inhibitor. It inhibited the uncoupled electron flow on photosystem II partial reaction from water to dichlorophenol indophenol (DCPIP) and from water to sodium silicomolybdate. However, the uncoupled partial reaction from diphenylcarbazide to DCPIP and the uncoupled photosystem I from DCPIPred to MV were not inhibited by 2.
CONCLUSIONS:
These results were corroborated by fluorescence decay data. Therefore, pachypodol inhibits the water-splitting enzyme activity. Compound 1 with a 4'-methoxy group was a weak inhibitor, indicating that the 4' free -OH group is important for strong inhibition.