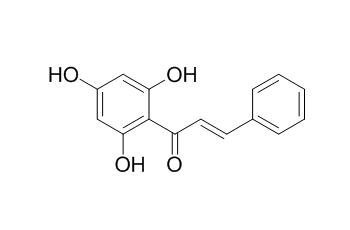
Pinocembrin chalcone
Pinocembrin chalcone has tyrosinase inhibitory activity.It also shows antimutagenic effect, which is mainly due to the inhibition of the first step of enzymatic activation of heterocyclic amines. Pinocembrin chalcone shows antimicrobial activity against the antibiotic susceptible NG strain WHO V ; it also displays activity against Candida albicans with a minimal inhibitory concentration value of 100 microg/mL.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Foods.2023, 12(6):1130.
Proc Biol Sci.2024, 291(2015):20232578.
J Ethnopharmacol.2017, 198:91-97
Crystals2020, 10(3), 206.
Evid Based Complement Alternat Med.2016, 2016:1230294
Life (Basel).2021, 11(12):1399.
Ecol Evol.2022, 12(11):e9459.
Korean Herb. Med. Inf.2021, 9(2):231-239.
Int J Mol Sci.2020, 21(19):7209.
Arch Toxicol.2017, 91(10):3225-3245
Related and Featured Products
Sex Transm Dis. 2011 Feb;38(2):82-8
Antimicrobial activity of flavonoids from Piper lanceaefolium and other Colombian medicinal plants against antibiotic susceptible and resistant strains of Neisseria gonorrhoeae.[Pubmed:
20921932 ]
The successful treatment of Neisseria gonorrhoeae (NG) infections is increasingly problematic because of the resistance of this pathogen to multiple antimicrobial agents. This development underscores the need for new antimicrobial sources. In the current study, 21 crude methanol extracts, from 19 plants used in Colombian traditional medicine for cutaneous infections, were screened for antimicrobial activity against NG.
METHODS AND RESULTS:
Extracts were screened by disc susceptibility assay. In addition, the minimum inhibitory concentrations of active compounds from P. lanceaefolium were assayed using a panel of 26 NG strains comprising 12 antibiotic-resistant phenotypes.
In all, 71% of the crude extracts exhibited antibacterial activity against the antibiotic susceptible NG strain WHO V, whereas 10% of the extracts inhibited penicillinase-producing NG strain GC1-182. The crude extract of Piper lanceaefolium was the only extract to show significant activity without ultraviolet (UV) light activation. Preliminary screening identified 3 compounds in this plant possessing antimicrobial activity: the flavonoids 5,7-dihydroxyflavanone (pinocembrin), 2',4',6'-trihydroxychalcone (Pinocembrin chalcone), and the prenylated benzoic acid derivative cyclolanceaefolic acid methyl ester. Pinocembrin and Pinocembrin chalcone inhibited 100% of the NG panel at 64 μg/mL and 128 μg/mL, respectively, whereas cyclolanceaefolic acid methyl ester inhibited 44% of the strains at 128 μg/mL.
CONCLUSIONS:
This is the first report of the antibacterial activity of Columbian plants against NG. The activity of the 2 flavonoids, pinocembrin, and Pinocembrin chalcone, toward both susceptible and resistant NG strains makes them promising candidates for further research.
Bioorg Med Chem. 2007 Mar 15;15(6):2396-402.
Synthesis and evaluation of 2',4',6'-trihydroxychalcones as a new class of tyrosinase inhibitors.[Pubmed:
17267225 ]
METHODS AND RESULTS:
Bioactivity-guided fractionation of a methanol extract from the leaves of Piper lanceaefolium resulted in the isolation of four new benzoic acid derivatives (1-4), together with taboganic acid, pinocembrin, and Pinocembrin chalcone.
CONCLUSIONS:
Lanceaefolic acid methyl ester (3) and Pinocembrin chalcone displayed activity against Candida albicans with a minimal inhibitory concentration value of 100 microg/mL in both cases.
J Agric Food Chem. 2001 Jun;49(6):3046-50.
Structural analysis of a novel antimutagenic compound, 4-Hydroxypanduratin A, and the antimutagenic activity of flavonoids in a Thai spice, fingerroot (Boesenbergia pandurata Schult.) against mutagenic heterocyclic amines.[Pubmed:
11410007]
METHODS AND RESULTS:
Six compounds were isolated from fresh rhizomes of fingerroot (Boesenbergia pandurata Schult.) as strong antimutagens toward 3-amino-1,4-dimethyl-5H-pyrido[4,3-b]indole (Trp-P-1) in Salmonella typhimurium TA98. These compounds were 2',4',6'-trihydroxychalcone (Pinocembrin chalcone; 1), 2',4'-dihydroxy-6'-methoxychalcone (cardamonin; 2), 5,7-dihydroxyflavanone (pinocembrin; 3), 5-hydroxy-7-methoxyflavanone (pinostrobin; 4), (2,4,6-trihydroxyphenyl)-[3'-methyl-2'-(3' '-methylbut-2' '-enyl)-6'-phenylcyclohex-3'-enyl]methanone (5), and (2,6-dihydroxy-4-methoxyphenyl)-[3'-methyl-2'-(3' '-methylbut-2' '-enyl)-6'-phenylcyclohex-3'-enyl]methanone (panduratin A; 6). Compound 5 was a novel compound (tentatively termed 4-hydroxypanduratin A), and 1 was not previously reported in this plant, whereas 2-4 and 6 were known compounds.
CONCLUSIONS:
The antimutagenic IC(50) values of compounds 1-6 were 5.2 +/- 0.4, 5.9 +/- 0.7, 6.9 +/- 0.8, 5.3 +/- 1.0, 12.7 +/- 0.7, and 12.1 +/- 0.8 microM in the preincubation mixture, respectively. They also similarly inhibited the mutagenicity of 3-amino-1-methyl-5H-pyrido[4,3-b]indole (Trp-P-2) and 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP). All of them strongly inhibited the N-hydroxylation of Trp-P-2. Thus, the antimutagenic effect of compounds 1-6 was mainly due to the inhibition of the first step of enzymatic activation of heterocyclic amines.
J Nat Prod. 2002 Jan;65(1):62-4.
Antifungal activity of benzoic acid derivatives from Piper lanceaefolium.[Pubmed:
11809068]
The aim of this study was to determine the antifungal activity of Drimenol (1) and its synthetic derivatives, nordrimenone (2), drimenyl acetate (3), and drimenyl-epoxy-acetate (4), and to establish a possible mechanism of action for Drimenol.
METHODS AND RESULTS:
For that, the effect of each compound on mycelial growth of Botrytis cinerea was assessed. Our results showed that compounds 1, 2, 3 and 4 are able to affect Botrytis cinerea growth with EC50 values of 80, 92, 80 and 314ppm, respectively.
CONCLUSIONS:
These values suggest that the activity of these compounds is mainly determined by presence of the double bond between carbons 7 and 8 of the drimane ring. In addition, germination of B. cinerea in presence of 40 and 80ppm of Drimenol is reduced almost to a half of the control value. Finally, in order to elucidate a possible mechanism by which Drimenol is affecting B. cinerea, the determination of membrane integrity, reactive oxygen species production and gene expression studies of specific genes were performed.
Biosci Biotechnol Biochem. 2009 Jul;73(7):1679-82.
Pinostrobin from Boesenbergia pandurata is an inhibitor of Ca2+-signal-mediated cell-cycle regulation in the yeast Saccharomyces cerevisiae.[Pubmed:
19584530]
METHODS AND RESULTS:
In this study, we synthesized a series of hydroxychalcones and examined their tyrosinase inhibitory activity. The results showed that 2',4',6'-trihydroxychalcone (Pinocembrin chalcone ,1), 2,2',3,4',6'-pentahydroxychalcone (4), 2',3,4,4',5,6'-hexahydroxychalcone (5), 2',4',6'-trihydroxy- 3,4-dimethoxychalcone (9) and 2,2',4,4',6'-pentahydroxychalcone (15) exhibited high inhibitory effects on tyrosinase with respect to l-tyrosine as a substrate.
CONCLUSIONS:
By the structure-activity relationship study, it was suggested that the 2',4',6'-trihydroxyl substructure in the chalcone skeleton were efficacious for the inhibition of tyrosinase activity. And also, the catechol structure on B-ring of chalcones was not advantageous for the inhibitory potency. Furthermore, 15 (IC(50)=1microM) was found to show the highest activity out of a set of 15 hydroxychalcones, even better than both 2,2',4,4'-tetrahydroxychalcone (13, IC(50)=5microM) and kojic acid (16, IC(50)=12microM), which were known as potent tyrosinase inhibitors. Kinetic study revealed that 15 acts as a competitive inhibitor of tyrosinase with K(i) value of 3.1microM.