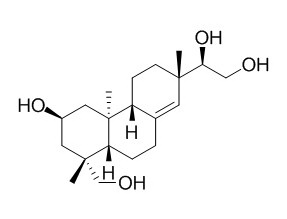
Kirenol
Kirenol possesses anti-bacteria, immunosuppression, anti-obesity, anti-oxidant, anti-inflammatory, anti-allergic, and anti-arthritic activities. Kirenol has significant potential for its discovery as a new lead compound for management of topical pain and inflammation; it can upregulate nuclear Annexin-1 which interacts with NF-κB to attenuate synovial inflammation of collagen-induced arthritis in rats. Kirenol can attenuate experimental autoimmune encephalomyelitis by inhibiting differentiation of Th1 and th17 cells and inducing apoptosis of effector T cells. Kirenol activates the BMP and Wnt/β-catenin signaling pathways.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Adv Healthc Mater.2024, 13(13):e2303276.
J of the Society of Cosmetic Scientists of Korea2018, 44(4):407-417
J. Soc. Cosmet. Sci. Korea2021, 47(1):57-63
J of Archaeological Science:Reports2024, 53:104298
Int J Mol Sci.2019, 20(16):E4015
Korean J. Medicinal Crop Sci.2022, 30(2):117-123.
Mol Med Rep.2022, 26(4):299.
Molecules.2021, 26(2):313.
Journal of Ginseng Research2022, j.jgr.2022.09.005.
Sci Rep.2015, 5:13194
Related and Featured Products
Sci Rep. 2015 Mar 12;5:9022.
Kirenol attenuates experimental autoimmune encephalomyelitis by inhibiting differentiation of Th1 and th17 cells and inducing apoptosis of effector T cells.[Pubmed:
25762107]
Experimental autoimmune encephalomyelitis (EAE), a model of multiple sclerosis (MS), is characterized by CNS demyelination mediated by autoreactive T cells. Kirenol, a biologically active substance isolated from Herba Siegesbeckiae, has potent anti-inflammatory activities.
METHODS AND RESULTS:
Here we investigated effects of Kirenol on EAE. Kirenol treatment markedly delayed onset of disease and reduced clinical scores in EAE mice. Kirenol treatment reduced expression of IFN-γ and IL-17A in the serum and proportion of Th1 and Th17 cells in draining lymph nodes. Priming of lymphocytes was reduced and apoptosis of MOG-activated CD4+ T cells was increased in Kirenol treated EAE mice. Kirenol treatment of healthy animals did not affect the lymphocytes in these non-immunized mice. Further in vitro studies showed that Kirenol inhibited viability of MOG-specific lymphocytes and induced apoptosis of MOG-specific CD4+ T cells in a dose- and time-dependent manner. Kirenol treatment upregulated Bax,downregulated Bcl-2,and increased activation of caspase-3 and release of cytochrome c, indicating that a mitochondrial pathway was involved in Kirenol induced apoptosis. Moreover, pretreatment with either a pan-caspase inhibitor z-VAD-fmk or a more specific caspase 3 inhibitor Ac-DEVD-CHO in lymphocytes reduced Kirenol induced apoptosis.
CONCLUSIONS:
Our findings implicate Kirenol as a useful agent for the treatment of MS.
Pharmacogn Mag. 2012 Apr;8(30):149-55.
Kirenol production in hairy root culture of Siegesbeckea orientalis and its antimicrobial activity.[Pubmed:
22701289]
Despite the excellent anti-inflammatory and anti-rheumatic efficacy associated with Kirenol generation, the content of Kirenol in Siegesbeckea orientalis is quite low.
This study was designed to establish a reliable Kirenol production protocol by transformed root cultures of S. orientalis and to investigate the antimicrobial activities of Kirenol, hairy root, and S. orientalis.
METHODS AND RESULTS:
Transformed root cultures of S. orientalis were established by the transformation of Agrobacterium rhizogenes A4. Transgenic status of the roots was confirmed by polymerase chain reaction (PCR) using rolB specific primers. The biomass and Kirenol accumulation of hairy root clones were assessed using four different culture media: MS, MS/2, B5, and white. The antimicrobial activities of Kirenol, hairy root, and S. orientalis were evaluated by the disc diffusion method.
The optimum media for Kirenol synthesis was MS. The content of Kirenol in transformed hairy roots made up about 80% of that observed in natural leaves of S. orientalis (1.6 mg/g dry weight). All tested samples displayed antimicrobial activity against Gram-positive pathogens including Staphylococcus epidermidis, Staphylococcus aureus, and Acinetobacter baumannii, with MIC ranging from 78 to 625 μg/mL.
CONCLUSIONS:
The high level of Kirenol contents was obtained from hairy roots of S. orientalis. Kirenol was effective against gram-positive bacteria. Interestingly, the extract from hairy roots showed a diverse antimicrobial effect from that of Kirenol and S. orientalis.
J Ethnopharmacol. 2011 Oct 11;137(3):1089-94.
Topical anti-inflammatory and analgesic activity of kirenol isolated from Siegesbeckia orientalis.[Pubmed:
21798328 ]
Siegesbeckia orientalis has been traditionally used as a topical anti-inflammatory and analgesic agent.
Current study was designed to explore the topical anti-inflammatory and analgesic effects of a constituent isolated from Siegesbeckia orientalis (Compositae), in order to validate its folk use.
METHODS AND RESULTS:
Kirenol was isolated from ethanolic extract of Siegesbeckia orientalis. Several topical formulations containing Kirenol were investigated for anti-inflammatory and analgesic activities in rat. The effects were studied using carrageenan-induced rat acute inflammation model, complete Freund's adjuvant (CFA)-induced chronic inflammation and formalin test in rats. Piroxicam gel and methyl salicylate ointment were studied as positive control for anti-inflammatory and analgesic activity, respectively.
The anti-inflammatory effect of Kirenol 0.4-0.5% (w/w) was similar to the effect of piroxicam gel 4h after carrageenan injection. The analgesic activity of topical preparation with more than 0.4% (w/w) was observed in the late phase. These effects may be due, at least in part, to the pro-inflammatory cytokine production of IL-1β and TNF-α. The administration of Kirenol cream at the dose of 0.3, 0.4 and 0.5% (w/w) significantly inhibited the development of joint swelling induced by CFA, which was auxiliary supported by histopathological studies.
CONCLUSIONS:
Kirenol has demonstrated its significant potential to be further investigated for its discovery as a new lead compound for management of topical pain and inflammation, although further pharmacological research is necessary to fully understand its mechanism of action. It also supports the potential beneficial effect of topically administered Siegesbeckia orientalis in inflammatory diseases.
Phytomedicine. 2012 Jul 15;19(10):882-9.
Kirenol exerts a potent anti-arthritic effect in collagen-induced arthritis by modifying the T cells balance.[Pubmed:
22673798 ]
Rheumatoid arthritis is characterized by the imbalance of T cells, which leads to increased pro-inflammatory and reduced anti-inflammatory cytokines. Modulating the balance among T cells is crucial for the treatment of RA.
Kirenol is a major diterpenoid components of Herba Siegesbeckiae, which has been applied for arthritic therapy for centuries. Since prior research showed Kirenol exhibited anti-inflammatory effect in rats, in this study we have evaluated the effect and mechanism of bioactive Kirenol in a rat model of collagen-induced arthritis (CIA) on modulation of T cells.
METHODS AND RESULTS:
After immunization with bovine type II collagen (CII), Wistar rats were orally administered saline (CIA group), 2 mg/kg Kirenol or 2 mg/kg prednisolone daily for 30 days. The severity of arthritis was clinically and histologically assessed. The numbers of CD4⁺CD25⁺Foxp3⁺ T regulatory cells (Tregs) and IFNγ⁺CD4⁺ and IL4⁺CD4⁺ T cells were determined by flow cytometry, the mRNA expression level of Foxp3 was quantified by RT-PCR, cytokine levels were measured by ELISA and CII-induced cell proliferation was quantified in vitro. Kirenol significantly delayed the occurrence and reduced the disease severity of CIA. Histological analysis confirmed Kirenol suppressed joint inflammation and inhibited cartilage and bone destruction, compared to the CIA group. Kirenol also upregulated the mRNA expression of Foxp3, increased the numbers of CD4⁺CD25⁺Foxp3⁺ and IL4⁺CD4⁺ T cells, and reduced the number of IFNγ⁺CD4⁺ T cells. Kirenol reduced the levels of TNF-α, IL-17A and IL-6 in synovial fluid and TNF-α, IL-17A and IFN-γ in serum, and increased the serum levels of IL-4, IL-10 and TGF-β1. In addition, Kirenol inhibited the ability of CII to induce splenocyte, PBMC and lymph node cell proliferation in vitro, compared to cells from CIA rats.
CONCLUSIONS:
In conclusion, these results suggest that Kirenol may be a potential immunosuppressant for the treatment for rheumatoid arthritis.
Biochem Biophys Res Commun. 2014 Mar 7;445(2):433-8.
Kirenol inhibits adipogenesis through activation of the Wnt/β-catenin signaling pathway in 3T3-L1 adipocytes.[Pubmed:
24530909]
Kirenol, a natural diterpenoid compound, has been reported to possess anti-oxidant, anti-inflammatory, anti-allergic, and anti-arthritic activities; however, its anti-adipogenic effect remains to be studied. The present study evaluated the effect of Kirenol on anti-adipogenesis through the activation of the Wnt/β-catenin signaling pathway.
METHODS AND RESULTS:
Kirenol prevented intracellular lipid accumulation by down-regulating key adipogenesis transcription factors [peroxisome proliferator-activated receptor γ (PPARγ), CCAAT/enhancer binding proteins α (C/EBPα), and sterol regulatory element binding protein-1c (SREBP-1c)] and lipid biosynthesis-related enzymes [fatty acid synthase (FAS) and acetyl-CoA carboxylase (ACC)], as well as adipocytokines (adiponectin and leptin). Kirenol effectively activated the Wnt/β-catenin signaling pathway, in which Kirenol up-regulated the expression of low density lipoprotein receptor related protein 6 (LRP6), disheveled 2 (DVL2), β-catenin, and cyclin D1 (CCND1), while it inactivated glycogen synthase kinase 3β (GSK3β) by increasing its phosphorylation. Kirenol down-regulated the expression levels of PPARγ and C/EBPα, which were up-regulated by siRNA knockdown of β-catenin.
CONCLUSIONS:
Overall, Kirenol is capable of inhibiting the differentiation and lipogenesis of 3T3-L1 adipocytes through the activation of the Wnt/β-catenin signaling pathway, suggesting its potential as natural anti-obesity agent.
Fitoterapia. 2014 Oct;98:59-65.
Kirenol stimulates osteoblast differentiation through activation of the BMP and Wnt/β-catenin signaling pathways in MC3T3-E1 cells.[Pubmed:
25062891]
Kirenol has been reported to possess anti-oxidant, anti-inflammatory, anti-allergic, anti-adipogenic, and anti-arthritic activities; however, its effect on osteoblast differentiation has not yet been reported. The aim of the present study was to evaluate the effect of Kirenol on osteoblast differentiation through activation of the bone morphogenetic protein (BMP) and Wnt/β-catenin signaling pathways in MC3T3-E1 cells.
METHODS AND RESULTS:
Kirenol markedly promoted alkaline phosphatase (ALP) activity and mineralization. Kirenol not only increased the expression of osteoblast differentiation markers, such as ALP, type I collagen (ColA1), and osteopontin (OPN), but also increased the expression of osteoprotegerin/receptor activator of nuclear factor kappa B ligand (OPG/RANKL) ratio. The effects of Kirenol on osteoblast differentiation were accompanied by stimulating the expression of the BMP and Wnt/β-catenin signaling pathways, including BMP2, runt-related transcription factor 2 (Runx2), osterix (Osx), low density lipoprotein receptor related protein 5 (LRP5), disheveled 2 (DVL2), β-catenin, cyclin D1 (CCND1), and phosphorylated glycogen synthase kinase 3β (GSK3β). In addition, Kirenol up-regulated the expression of β-catenin, CCND1, ALP, and ColA1 which were down-regulated by siRNA knockdown of β-catenin.
CONCLUSIONS:
Overall, these results demonstrate that Kirenol is capable of promoting osteoblast differentiation in MC3T3-E1 cells through activation of the BMP and Wnt/β-catenin signaling pathways, suggesting that it is a potential candidate target for treating or preventing osteoporosis.
Pharmazie. 2014 Feb;69(2):148-53.
Kirenol, a compound from Herba Siegesbeckiae, induces apoptosis in human chronic myeloid leukemia K562 cells.[Pubmed:
24640606]
Kirenol is a biologically active substance isolated from Herba Siegesbeckiae.
METHODS AND RESULTS:
In the experiments, we explored a novel antitumor activity of Kirenol. The data demonstrated that Kirenol had strong cytotoxic effects to human chronic myeloid leukemia K562 cells, the 50% inhibitory concentration (IC50) for Kirenol was 53.05 ig/ml, 18.19 pg/ml and 15.08 microg/ml for 24, 48 and 72 h, determined using the MTT assay. Further studies showed that Kirenol treatment caused externalization of phosphatidylserine, accumulation of ROS (reactive oxygen species), alteration of mitochondrial membrane potential, release of cytochrome c, reduction in the level of the Bcl-2 protein and upregulation of Bax and tBid, Kirenol induced cell apoptosis in a caspase-independent manner. Further studies indicated that Kirenol treatment triggered the arrest of cell cycle S period which might resulted from the up-regulation of phosphorylation of p53 (Ser 6 and Ser 37) and expression of p21 protein.
CONCLUSIONS:
Our results indicated that Kirenol possesses antitumor action on human chronic myeloid leukemia K562 cells in vitro. Kirenol may have therapeutic potential for the treatment of cancer that deserves further investigation.
J Ethnopharmacol. 2011 Sep 1;137(1):774-82.
Kirenol upregulates nuclear annexin-1 which interacts with NF-κB to attenuate synovial inflammation of collagen-induced arthritis in rats.[Pubmed:
21745559 ]
Kirenol is a diterpenoid compound purified from the Chinese Herba Siegesbeckiae. Siegesbeckiae has been employed for the treatment of arthritis for centuries, its safety and efficacy are documented through a long history of human use.
To investigate the effects on collagen-induced arthritis (CIA) and anti-inflammatory mechanism of Kirenol.
METHODS AND RESULTS:
Kirenol was administrated intragastrically in rats after the onset of CIA. Pathological changes were evaluated by paw swelling and histopathology. Concentration of IL-1β in synovial fluid and adrenal corticotropin (ACTH) in plasma were determined by Elisa. Western blot was performed to detect the expression of annexin-1 and glucocorticoid receptor alpha (GRα) in synovium. NF-κB DNA binding activity was assessed by electrophoretic mobility shift assays (EMSA).
Kirenol (1, 2, and 4 mg/kg) and prednisolone depressed paw swelling and reduced IL-1β of synovial fluid in the CIA rats (p<0.05 or p<0.01). Kirenol and prednisolone upregulated nuclear annexin-1 and inhibited NF-κB activity in synovium of CIA. The inhibitory effect of Kirenol and prednisolone on NF-κB activity was enhanced by anti-annexin-1 Ab. Prednisolone, but not Kirenol, downregulated plasma ACTH and GRα expression significantly (p<0.01).
CONCLUSIONS:
Kirenol and prednisolone can upregulate nuclear annexin-1 which interacts with NF-κB to inhibit NF-κB activity, reduce cytokines expression and thereby attenuate inflammation of CIA joints. Kirenol does not lead to ACTH or GR downregulation, which is in contrast to classic glucocorticoid prednisolone. Kirenol shares with GCs similar anti-inflammatory mechanism but bypass the considerable limitation of GCs treatment.