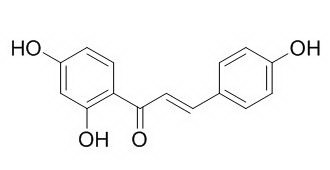
Isoliquiritigenin
Isoliquiritigenin has hepatoprotective, chemopreventive, antitumor, vasorelaxant, anti-platelet, anti-allergic, antiviral, antioxidant and anti-inflammatory effects, it can induce growth inhibition and apoptosis through downregulating AA metabolic network and the deactivation of PI3K/Akt in human breast cancer. Isoliquiritigenin also has the ability to protect cells from AA+iron-induced H2O2 production and mitochondrial dysfunction, which is mediated with GSK3β phosphorylation downstream of AMPK.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Cosmetics2025, 12(3), 108
Heliyon.2024, 10(11):e32352.
Front Cell Dev Biol.2020, 8:32.
ACS Food Sci. Technol.2023, 3(2):273-282.
Vietnam J. Chem.2023, 61(3),308-317
Medicina (Kaunas).2020, 56(12):685.
Oxid Med Cell Longev.2022, 2022:9139338.
LWT2020, 124:109163
Front. Pharmacol.2022, 901563.
Biol Pharm Bull.2018, 41(11):1685-1693
Related and Featured Products
J Ethnopharmacol. 2015 Mar 13;162:134-9.
A protective mechanism of licorice (Glycyrrhiza uralensis): isoliquiritigenin stimulates detoxification system via Nrf2 activation.[Pubmed:
25557030]
Licorice (Glycyrrhizae radix), the root of Glycyrrhiza uralensis Fisch. (Leguminosae), is mainly used to moderate the characteristics of toxic herbs in Traditional Chinese Medicine, which could be partly interpreted as detoxification. However, the underlying mechanism is still not fully elucidated. Nuclear factor erythroid 2-related factor 2 (Nrf2) plays a key role in the protection against toxic xenobiotics. In our previous research, we have identified that extracts from Glycyrrhiza uralensis induced the expression of Nrf2 nuclear protein and its downstream genes. This research aims to screen the most potent Nrf2 inducer isolated from Glycyrrhiza uralensis and examine its effect on Nrf2 signaling pathway and detoxification system.
METHODS AND RESULTS:
Four compounds derived from Glycyrrhiza uralensis (glycyrrhetinic acid, liquiritigenin, Isoliquiritigenin and liquiritin) were screened by ARE-luciferase reporter. The most potent ARE-luciferase inducer was chosen to further examine its effect on Nrf2 and detoxification genes in HepG2 cells. The role of Nrf2-dependent mechanism was tested by using Nrf2 knockout mice (Nrf2 KO) and Nrf2 wild-type mice (Nrf2 WT).
ARE-luciferase reporter assay showed these four compounds were all potent Nrf2 inducers, and Isoliquiritigenin was the most potent inducer. Isoliquiritigenin significantly up-regulated the expression of Nrf2 and its downstream detoxification genes UDP-glucuronosyltransferase 1A1 (UGT1A1), glutamate cysteine ligase (GCL), multidrug resistance protein 2 (MRP2) and bile salt export pump (BSEP) in vitro and in vivo. Additionally, Isoliquiritigenin showed Nrf2-dependent transactivation of UGT1A1, GCLC and MRP2.
CONCLUSIONS:
Isoliquiritigenin, isolated from Glycyrrhiza uralensis, stimulates detoxification system via Nrf2 activation, which could be a potential protective mechanism of licorice.
Eur J Pharmacol. 2008 Apr 14;584(1):175-84.
Isoliquiritigenin isolated from the roots of Glycyrrhiza uralensis inhibits LPS-induced iNOS and COX-2 expression via the attenuation of NF-kappaB in RAW 264.7 macrophages.[Pubmed:
18295200]
In this study, the anti-inflammatory effects of flavonoids isolated from the roots of Glycyrrhiza uralensis (Leguminosae), namely, isoliquiritin (the glycoside of isoliquirigenin) and Isoliquiritigenin (the aglycone of isoliquiritin) were evaluated on lipopolysaccharide (LPS)-treated RAW 264.7 macrophages.
METHODS AND RESULTS:
Isoliquiritigenin (ILG) more potently inhibited LPS-induced nitric oxide (NO) and prostaglandin E(2) (PGE(2)) production than isoliquiritin (ILT). Consistent with these findings, ILG reduced the LPS-induced expressions of inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2) at the protein and mRNA levels in a concentration-dependent manner, as determined by Western blotting and RT-PCR, respectively. In addition, the release of tumor necrosis factor-alpha (TNF-alpha) and interleukin-6 (IL-6), and the mRNA expression levels of these cytokines were reduced by ILG in a dose-dependent manner. Moreover, ILG attenuated the LPS-induced DNA binding activity and the transcription activity of nuclear factor-kappa B (NF-kappaB), and this was associated with a decrease in inhibitory kappa B-alpha (IkappaB-alpha) phosphorylation and in the subsequent blocking of p65 and p50 protein translocations to the nucleus. Furthermore, ILG suppressed the phosphorylations of IkappaB kinase (IKK), ERK1/2, and p38, whereas the phosphorylation of JNK1/2 was unaffected.
CONCLUSIONS:
These results suggest that the anti-inflammatory properties of ILG are caused by iNOS, COX-2, TNF-alpha, and IL-6 down-regulation due to NF-kappaB inhibition via the suppression of IKK, ERK1/2 and p38 phosphorylation in RAW 264.7 cells.
Biochem Pharmacol. 2010 May 1;79(9):1352-62.
AMPK-mediated GSK3beta inhibition by isoliquiritigenin contributes to protecting mitochondria against iron-catalyzed oxidative stress.[Pubmed:
20026081 ]
Isoliquiritigenin (ILQ), a flavonoid compound originated from Glycyrrhiza species, is known to activate SIRT1. Arachidonic acid (AA) in combination with iron (a catalyst of auto-oxidation) leads cells to produce excess reactive species with a change in mitochondrial permeability transition.
METHODS AND RESULTS:
In view of the importance of oxidative stress in cell death and inflammation, this study investigated the potential of ILQ to protect cells against the mitochondrial impairment induced by AA+iron and the underlying basis for this cytoprotection. Treatment with ILQ inhibited apoptosis induced by AA+iron, as evidenced by alterations in the levels of the proteins associated with cell viability: ILQ prevented a decrease in Bcl-x(L), and cleavage of poly(ADP-ribose)polymerase and procaspase-3. Moreover, ILQ inhibited the ability of AA+iron to elicit mitochondrial dysfunction. In addition, superoxide generation in mitochondria was attenuated by ILQ treatment. Consistently, ILQ prevented cellular H2O2 production increased by AA+iron, thereby enabling cells to restore GSH content. ILQ treatment enhanced inhibitory phosphorylation of glycogen synthase kinase-3beta (GSK3beta), and prevented a decrease in the GSK3beta phosphorylation elicited by AA+iron, which contributed to protecting cells and mitochondria. GSK3beta phosphorylation by ILQ was preceded by AMP-activated protein kinase (AMPK) activation, which was also responsible for mitochondrial protection, as shown by reversal of its effect in the experiments using a dominant negative mutant of AMPK and compound C. Moreover, the AMPK activation led to GSK3beta phosphorylation.
CONCLUSIONS:
These results demonstrate that ILQ has the ability to protect cells from AA+iron-induced H2O2 production and mitochondrial dysfunction, which is mediated with GSK3beta phosphorylation downstream of AMPK.
Planta Med. 2015 May;81(7):586-93.
In Vivo Gastroprotective Effect along with Pharmacokinetics, Tissue Distribution and Metabolism of Isoliquiritigenin in Mice.[Pubmed:
25875506]
As numerous herbal products have been used as dietary supplements or functional foods, the demands of the pharmacokinetic and pharmacodynamic characteristics of active compounds are increasing in order to secure a consistent outcome (i.e., efficiency and safety).
METHODS AND RESULTS:
In this study, the pharmacokinetics including tissue distribution, metabolism, and protein binding of Isoliquiritigenin, a chalcone found in Glycyrrhiza glabra, and its metabolite, liquiritigenin, at various doses in mice are reported. Also, correlations between the preferential tissue distribution and pharmacological effect of Isoliquiritigenin in certain organs were investigated using the in vivo gastroprotective effect of Isoliquiritigenin in mice with indomethacin-induced ulcer. The absorbed fraction of Isoliquiritigenin was high, but the absolute bioavailability was low mainly due to its metabolism. In spite of the low bioavailability, the gastroprotective effect of Isoliquiritigenin was attributed to its high distribution in the stomach. Isoliquiritigenin prevented the occurrence of gastric ulcers by indomethacin, which is associated with increased gastric mucous secretion because the pretreatment with Isoliquiritigenin presumably counteracted the decreased cyclooxygenase 2 by indomethacin.
CONCLUSIONS:
This may suggest that the pharmacokinetic properties of Isoliquiritigenin are useful to predict its efficacy as a gastroprotective agent in a target organ such as the stomach.
Toxicol Appl Pharmacol. 2013 Oct 1;272(1):37-48.
Isoliquiritigenin induces growth inhibition and apoptosis through downregulating arachidonic acid metabolic network and the deactivation of PI3K/Akt in human breast cancer.[Pubmed:
23747687]
Arachidonic acid (AA)-derived eicosanoids and its downstream pathways have been demonstrated to play crucial roles in growth control of breast cancer.
METHODS AND RESULTS:
Here, we demonstrate that Isoliquiritigenin, a flavonoid phytoestrogen from licorice, induces growth inhibition and apoptosis through downregulating multiple key enzymes in AA metabolic network and the deactivation of PI3K/Akt in human breast cancer. Isoliquiritigenin diminished cell viability, 5-bromo-2'-deoxyuridine (BrdU) incorporation, and clonogenic ability in both MCF-7 and MDA-MB-231cells, and induced apoptosis as evidenced by an analysis of cytoplasmic histone-associated DNA fragmentation, flow cytometry and hoechst staining. In addition, it downregulated the levels of phospho-PI3K, phospho-PDK (Ser(241)), phospho-Akt (Thr(308)), phospho-Bad (Ser(136)), and Bcl-xL expression, thereby activating caspase cascades and eventually cleaving poly(ADP-ribose) polymerase (PARP). Conversely, the addition of exogenous eicosanoids, including PGE2, LTB4 and a 20-HETE analog (WIT003), and caspase inhibitors, or overexpression of constitutively active Akt reversed Isoliquiritigenin-induced apoptosis. Notably, Isoliquiritigenin induced growth inhibition and apoptosis of MDA-MB-231 human breast cancer xenografts in nude mice, together with decreased intratumoral levels of eicosanoids and phospho-Akt (Thr(308)).
CONCLUSIONS:
Collectively, these data suggest that Isoliquiritigenin induces growth inhibition and apoptosis through downregulating AA metabolic network and the deactivation of PI3K/Akt in human breast cancer.
Fitoterapia. 2013 Jan;84:208-12.
Isoliquiritigenin showed strong inhibitory effects towards multiple UDP-glucuronosyltransferase (UGT) isoform-catalyzed 4-methylumbelliferone (4-MU) glucuronidation.[Pubmed:
23237733]
Isoliquiritigenin, a herbal ingredient with chalcone structure, has been speculated to be able to inhibit one of the most drug-metabolizing enzymes (DMEs) UDP-glucuronosyltransferase (UGT).
METHODS AND RESULTS:
Therefore, the aim of the present study was to investigate the inhibition of Isoliquiritigenin towards important UGT isoforms in the liver and intestine, including UGT1A1, 1A3, 1A6, 1A7, 1A8, 1A9 and 1A10. The recombinant UGT-catalyzed 4-methylumbelliferone (4-MU) glucuronidation was used as probe reactions. The results showed that 100μM of Isoliquiritigenin inhibited the activity of UGT1A1, UGT1A3, UGT1A6, UGT1A7, UGT1A8, UGT1A9, and UGT1A10 by 95.2%, 76.1%, 78.9%, 87.2%, 67.2%, 94.8%, and 91.7%, respectively. The data fitting using Dixon plot and Lineweaver-Burk plot showed that the inhibition of UGT1A1, UGT1A9 and UGT1A10 by Isoliquiritigenin was all best fit to the competitive inhibition, and the second plot using the slopes from the Lineweaver-Burk plot versus Isoliquiritigenin concentrations was used to calculate the inhibition kinetic parameter (K(i)) to be 0.7μM, 0.3μM, and 18.3μM for UGT1A1, UGT1A9, and UGT1A10, respectively.
CONCLUSIONS:
All these results indicated the risk of clinical application of Isoliquiritigenin on the drug-drug interaction and other possible diseases induced by the inhibition of Isoliquiritigenin towards these UGT isoforms.
Chem Biol Interact. 2015 Jan 5;225:13-20.
Isoliquiritigenin attenuates oxidative hepatic damage induced by carbon tetrachloride with or without buthionine sulfoximine.[Pubmed:
25450236]
Glycyrrhizae radix (G. radix) has been demonstrated to have hepatoprotective properties. This study determined the therapeutic effects of Isoliquiritigenin (isoLQ) in G. radix, against liver injury induced by CCl4 in rats.
METHODS AND RESULTS:
CCl4 (0.5 ml/kg/d, twice) or CCl4 plus buthionine sulfoximine exerted severe liver damage assessed by increased plasma levels of alanine aminotransferase and aspartate aminotransferase, in addition to hepatic degeneration and necrosis. These pathological changes were markedly protected by pretreatment with isoLQ (5, 20 mg/kg/d, p.o.) for 3 consecutive days. In addition, pretreatment with isoLQ inhibited CCl4-induced reduction of cytochrome P450 2E1 protein and mRNA expression as well as activity in the liver. Moreover, isoLQ pretreatment reversed the decrease in hepatic antioxidant capacity induced by CCl4 as well as suppressed expression of tumor necrosis factor-alpha and cyclooxigenase-2 in the liver.
CONCLUSIONS:
These results suggest that isoLQ has a protective effect against CCl4-induced liver damage through induction of antioxidant and anti-inflammatory activities.
Pharmacol Res. 2006 Mar;53(3):303-9.
Protective effects of isoliquiritigenin in transient middle cerebral artery occlusion-induced focal cerebral ischemia in rats.[Pubmed:
16459097 ]
Epidemiological studies indicate that the intake of flavonoids is inversely associated with risk of stroke, cardiovascular diseases and cancer. Isoliquiritigenin (ISL), a flavonoid constituent in the root of Glycyrrhiza glabra, is known to have vasorelaxant effect, antioxidant, anti-platelet, anti-tumor, anti-allergic, antiviral activities and estrogenic properties. However, there is no report on the effects of ISL in cerebral ischemia. Evidence demonstrate that the impaired energy metabolism and the excessive generation of reactive oxygen radicals (ROS) contribute to the brain injury associated with cerebral ischemia.
METHODS AND RESULTS:
In the present study, the protective effects of ISL were investigated in transient middle cerebral artery occlusion (MCAO)-induced focal cerebral ischemia-reperfusion injury in rats. Male Sprague-Dawley rats were divided into five groups: sham-operated group, vehicle-pretreated group, and three ISL-pretreated groups (5, 10 and 20 mg kg(-1), i.g.). ISL were administered once a day, for 7 days prior to ischemia. The rats were subjected to 2 h right MCAO via the intraluminal filament technique and 22 h reperfusion. Pretreatment with ISL significantly reduced the cerebral infarct volume and edema and produced significant reduction in neurological deficits. In this study, in order to clarify the mechanism of ISL's protection against cerebral ischemia damage, cerebral energy metabolism, brain Na+K+ATPase activity, malondialdehyde (MDA) content and antioxidant enzyme activities were measured. ISL pretreatment increased the brain ATP content, energy charge (EC) and total adenine nucleotides (TAN) in a dose-dependent manner. The brain Na+K+ATPase activity was protected significantly by pretreatment of ISL for 7 days. Pretreatment with ISL significantly inhibited the increases of brain MDA content and prevented the activities of brain superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GSH-Px) from declines caused by cerebral ischemia-reperfusion.
CONCLUSIONS:
All these findings indicate that ISL has the protective potential against cerebral ischemia injury and its protective effects may be due to the amelioration of cerebral energy metabolism and its antioxidant property.