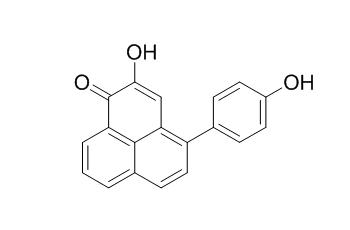
Irenolone
Irenolone is a natural product from Musa itinerans Cheesm.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Plants (Basel).2020, 9(11):1535.
Evid Based Complement Alternat Med.2019, 2019:2135351
Mol Med Rep.2015, 12(5):7789-95
Processes2021, 9(11),2065.
Planta Med.2023, a-2192-2281.
Cancer Lett. 2023, 18:216584.
Journal of Holistic Integrative Pharmacy2024, 5(1):45-55.
J Ethnopharmacol.2017, 209:305-316
Planta Med.2019, 85(9-10):766-773
Evid Based Complement Alternat Med.2020, 2020:8582318.
Related and Featured Products
Phytochemistry. 2014 Jul;103:171-177.
Diarylheptanoids and phenylphenalenones from Musa itinerans fruits.[Pubmed:
24766994]
METHODS AND RESULTS:
Two diarylheptanoids, musaitinerins A and B, one heterodimeric phenylphenalenone musaitinerone and four known phenylphenalenones, identified as 4-hydroxy-2-methoxy-9-phenyl-1H-phenalen-1-one, musanolone E, hydroxyanigorufone and Irenolone were isolated from the fruits of Musa itinerans Cheesm. Their structures were elucidated using spectroscopic analyses. The antimicrobial activity of these compounds was evaluated against Escherichia coli, Staphylococcus aureus and Candida albicans; the cytotoxic activity of these compounds was also evaluated against human erythromyeloblastoid leukemia (K562) and human alveolar carcinoma epithelial (A549) cell lines, respectively.
CONCLUSIONS:
Musaitinerone and musanolone E exhibited weak effects against the A549 cell line, as compared with adriamycin. However, these two compounds did not exhibit any growth inhibition against K562 cells, S. aureus, E. coli or C. albicans. The other compounds were inactive against all of the tested cell lines and microorganisms, even at concentrations as high as 50 μM.
Biosci Biotechnol Biochem. 2000 Oct;64(10):2089-98.
Changes in the content and biosynthesis of phytoalexins in banana fruit.[Pubmed:
11129580 ]
METHODS AND RESULTS:
Changes in the phytoalexin content in unripe fruit of banana, Musa acuminata, were analyzed after various treatments. The results show that level of hydroxyanigorufone started to increase 1-2 day after either wounding or inoculation with conidia of Colletotrichum musae. Inoculation followed by wounding induced the formation of many other phenylphenalenones. The accumulation of hydroxyanigorufone decreased, after its transient maximum, on ripening by exposure of the wounded fruit to ethylene. The level of production of hydroxyanigorufone in ripe fruit treated by wounding and/or by inoculation was much lower than that in unripe fruit. 2-Aminooxyacetic acid, an inhibitor of phenylalanine ammonia-lyase (PAL), inhibited the accumulation of hydroxyanigorufone in wounded fruit, and the PAL activity increased after wounding and ethylene treatment, respectively.
CONCLUSIONS:
Feeding experiments with [1-(13)C] and [2-(13)C]cinnamic acids, and [2-(13)C]malonate show that two molecules of cinnamic acid and one of malonate were incorporated into each molecule of hydroxyanigorufone. The phytoalexins isolated from fruit to which deuterated hydroxyanigorufone and Irenolone had been administered revealed that 2-(4'-hydroxyphenyl)-1,8-naphthalic anhydride was biosynthesized from hydroxyanigorufone rather than from Irenolone.