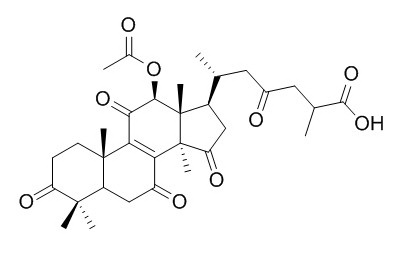
Ganoderic acid F
Ganoderic acid F has anti-hepatitis B, anti-inflammatory, and anti-tumor-promoting activities.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Separations2021, 8(6),80.
Evid Based Complement Alternat Med.2016, 2016:4357656
Heinrich Heine University Dusseldorf2021, 62203.
Korean J of Medicinal Crop Science2018, 220-226
Molecules.2019, 24(24):E4536
Nat Prod Commun.2018, 10.1177
Oncotarget.2017, 8(53):90925-90947
Phytomedicine.2022, 102:154183.
Talanta Open2023, 7:100227
Comparative Clinical Pathology 2021, 30:961-971.
Related and Featured Products
Methyl ganoderate F
Catalog No: CFN95584
CAS No: 98665-08-8
Price: $368/5mg
12beta-Acetoxy-7beta-hydroxy-3,11,15,23-tetraoxo-5alpha-lanosta-8,20-dien-26-oic acid
Catalog No: CFN95515
CAS No: 1245946-62-6
Price: $318/5mg
Ganoderenic acid K
Catalog No: CFN95469
CAS No: 942950-94-9
Price: $318/10mg
Ganoderic acid C6
Catalog No: CFN92290
CAS No: 105742-76-5
Price: $318/20mg
Methyl ganoderate C6
Catalog No: CFN92292
CAS No: 105742-81-2
Price: Inquiry(manager@chemfaces.com)
Ganoderic acid H
Catalog No: CFN92056
CAS No: 98665-19-1
Price: $318/10mg
Methyl ganoderate H
Catalog No: CFN92291
CAS No: 98665-11-3
Price: Inquiry(manager@chemfaces.com)
Ganoweberianic acid E
Catalog No: CFN95504
CAS No: 1309931-90-5
Price: $413/5mg
12beta-Acetoxy-3beta-hydroxy-7,11,15,23-tetraoxo-lanost-8,20-diene-26-oic acid
Catalog No: CFN95468
CAS No: 1085338-75-5
Price: $318/5mg
12beta-Acetoxy-3,7,11,15,23-pentaoxo-lanost-8,20-dien-26-oic acid
Catalog No: CFN95505
CAS No: 1309931-91-6
Price: $318/5mg
Evid Based Complement Alternat Med. 2012;2012:780892.
Pharmacokinetics of ganoderic acids a and f after oral administration of ling zhi preparation in healthy male volunteers.[Pubmed:
22577465]
The objectives of this paper were to evaluate the pharmacokinetics of ganoderic acid A and Ganoderic acid F after a single oral dose of the water extract of MG2-strain Ling Zhi (MG2FB-WE) and to assess the influence of food on the pharmacokinetics in 12 healthy male volunteers.
METHODS AND RESULTS:
This study was a single-dose, open-label, randomized, two-phase crossover study with at least 2 wk washout period. Each subject was randomly assigned to receive a single oral dose of 3,000 mg of MG2FB-WE in granular formulation dissolved in 200 mL of warm water, either under a fasting condition, or immediately after a standard breakfast (fed condition). Blood samples were collected immediately before and at specific time points until 8 h after MG2FB-WE administration. Plasma ganoderic acid A and Ganoderic acid F concentrations were determined by using liquid chromatography-mass spectrometry (LC-MS) technique.
CONCLUSIONS:
In conclusion, the pharmacokinetic profile of both ganoderic acids under a fasting condition was characterized by rapid absorption from the gastrointestinal tract (T(max) at approximately 30 min) and a short elimination half-life (<40 min). Food significantly decreased C(max) and delayed T(max), but did not affect the extent of ganoderic acid A absorption. However, concomitant food intake markedly impeded both rate and extent of Ganoderic acid F absorption.