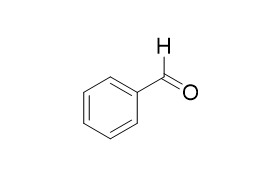
Benzaldehyde
Reference standards.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Antioxidants (Basel).2021, 10(10):1620.
Evid Based Complement Alternat Med.2021, 2021:6687513.
Proc Biol Sci.2024, 291:20232298.
J Korean Soc Food Sci Nutr2023, 52(7): 750-757
Int. J. Mol. Sci.2022, 23(14),7699;
J Plant Biotechnol.2023, 50:070-075.
Chem Biol Interact.2018, 290:44-51
Planta Med.2023, a-2192-2281.
Molecules.2019, 24(11):E2044
PLoS One.2021, 16(9):e0257243.
Related and Featured Products
Journal of the American Chemical Society, 2008, 130(18):5854-5.
Size-SelectiveLewis Acid Catalysis in a Microporous Metal-Organic Framework with Exposed Mn 2+ Coordination Sites.[Reference:
WebLink]
METHODS AND RESULTS:
Treatment of selected aldehydes and ketones with cyanotrimethylsilane in the presence of the microporous metal-organic framework Mn3[(Mn4Cl)3BTT8(CH3OH)10]2 (1, H3BTT = 1,3,5-benzenetristetrazol-5-yl) leads to rapid conversion to the corresponding cyanosilylated products. The transformation is catalyzed by coordinatively unsaturated Mn2+ ions that serve as Lewis acids and lead to conversion yields of 98 and 90% for Benzaldehyde and 1-naphthaldehyde, the highest thus far for a metal-organic framework. Larger carbonyl substrates cannot diffuse through the pores of 1, and conversion yields are much lower for these, attesting to the heterogeneity of the reaction and its dependence on guest size. The Mukaiyama-aldol reaction, known to require much more active Lewis catalysts, is also catalyzed in the presence of 1, representing the first such example for a metal-organic framework. Conversion yields obtained for the reaction of selected aldehydes with silyl enolates reach 63%, on par with those obtained with zeolites.
CONCLUSIONS:
Size selectivity is demonstrated for the first time with this reaction through the use of larger silyl enolate substrates.