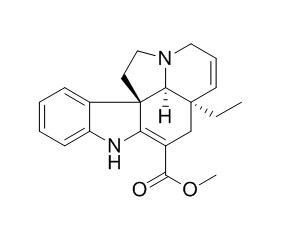
Tabersonine
Tabersonine is a precursor for vincristine used in cancer chemotherapy, the biocompatibility and small size essential for permeating the blood-brain barrier make it a potential therapeutic drug candidate for treating AD.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
J Agric Food Chem.2020, 68(51):15164-15175
Korean J Pain.2021, 34(4):405-416.
J Agric Food Chem.2021, 69(14):4210-4222.
Planta Med.2022, a-1876-3009.
Int J Mol Sci.2020, 21(19):7209.
J Agric Food Chem.2024, 72(45):25219-25228.
Nutrients.2018, 10(10)
Molecules.2022, 27(21):7643.
BMB Rep.2020, 53(4):218-222.
Front Pharmacol.2023, 14:1244655.
Related and Featured Products
ACS Chem Neurosci. 2015 Jun 17;6(6):879-88.
Tabersonine Inhibits Amyloid Fibril Formation and Cytotoxicity of Aβ(1-42).[Pubmed:
25874995 ]
The misfolding and aggregation of amyloid beta (Aβ) peptides into amyloid fibrils are key events in the amyloid cascade hypothesis for the etiology of Alzheimer's disease (AD).
METHODS AND RESULTS:
Using thioflavin-T (ThT) fluorescence assay, atomic force microscopy, circular dichroism, size exclusion chromatography, surface plasmon resonance (SPR), and cytotoxicity tests, we demonstrate that Tabersonine, an ingredient extracted from the bean of Voacanga africana, disrupts Aβ(1-42) aggregation and ameliorates Aβ aggregate-induced cytotoxicity. A small amount of Tabersonine (e.g., 10 μM) can effectively inhibit the formation of Aβ(1-42) (e.g., 80 μM) fibrils or convert mature fibrils into largely innocuous amorphous aggregates. SPR results indicate that Tabersonine binds to Aβ(1-42) oligomers in a dose-dependent way. Molecular dynamics (MD) simulations further confirm that Tabersonine can bind to oligomers such as the pentamer of Aβ(1-42). Tabersonine preferentially interact with the β-sheet grooves of Aβ(1-42) containing aromatic and hydrophobic residues. The various binding sites and modes explain the diverse inhibitory effects of Tabersonine on Aβ aggregation.
CONCLUSIONS:
Given that Tabersonine is a natural product and a precursor for vincristine used in cancer chemotherapy, the biocompatibility and small size essential for permeating the blood-brain barrier make it a potential therapeutic drug candidate for treating AD.
Phytochemistry. 2003 Sep;64(2):401-9.
Jasmonate-induced epoxidation of tabersonine by a cytochrome P-450 in hairy root cultures of Catharanthus roseus.[Pubmed:
12943756]
Methyl jasmonate, a chemical inducer of secondary metabolism, was shown to promote Tabersonine 2 biosynthesis in hairy root cultures of Catharanthus roseus.
METHODS AND RESULTS:
Tabersonine 6,7-epoxidase activity was detected in total protein extract of jasmonate-induced hairy root cultures using labeled 14C-Tabersonine 2. This enzyme converted Tabersonine 2 to lochnericine 3 by selective epoxidation at positions 6 and 7 via a reaction dependent on NADPH and molecular oxygen. Carbon monoxide, clotrimazole, miconazole, and cytochrome C were shown to be strong inhibitors of the enzyme.
CONCLUSIONS:
The activity was found in microsomes, indicating that Tabersonine 6,7-epoxidase was a cytochrome P-450-dependent monooxygenase.
Proc Natl Acad Sci U S A. 2015 May 12;112(19):6224-9.
Completion of the seven-step pathway from tabersonine to the anticancer drug precursor vindoline and its assembly in yeast.[Pubmed:
25918424 ]
Antitumor substances related to vinblastine and vincristine are exclusively found in the Catharanthus roseus (Madagascar periwinkle), a member of the Apocynaceae plant family, and continue to be extensively used in cancer chemotherapy. Although in high demand, these valuable compounds only accumulate in trace amounts in C. roseus leaves. Vinblastine and vincristine are condensed from the monoterpenoid indole alkaloid (MIA) precursors catharanthine and vindoline. Although catharanthine biosynthesis remains poorly characterized, the biosynthesis of vindoline from the MIA precursor Tabersonine is well understood at the molecular and biochemical levels.
METHODS AND RESULTS:
This study uses virus-induced gene silencing (VIGS) to identify a cytochrome P450 [CYP71D1V2; Tabersonine 3-oxygenase (T3O)] and an alcohol dehydrogenase [ADHL1; Tabersonine 3-reductase (T3R)] as candidate genes involved in the conversion of Tabersonine or 16-methoxyTabersonine to 3-hydroxy-2,3-dihydroTabersonine or 3-hydroxy-16-methoxy-2,3-dihydroTabersonine, which are intermediates in the vindorosine and vindoline pathways, respectively. Biochemical assays with recombinant enzymes confirm that product formation is only possible by the coupled action of T3O and T3R, as the reaction product of T3O is an epoxide that is not used as a substrate by T3R.
CONCLUSIONS:
The T3O and T3R transcripts were identified in a C. roseus database representing genes preferentially expressed in leaf epidermis and suggest that the subsequent reaction products are transported from the leaf epidermis to specialized leaf mesophyll idioblast and laticifer cells to complete the biosynthesis of these MIAs. With these two genes, the complete seven-gene pathway was engineered in yeast to produce vindoline from Tabersonine.