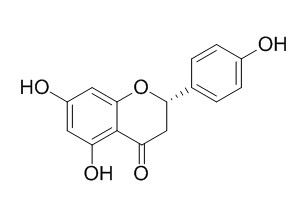
Naringenin
Naringenin is a weak estrogen that also exhibits partial antiestrogenic activity in the female rat uterus and MCF-7 human breast cancer cells. Naringenin is also a agent for the treatment of hepatitis C virus (HCV) infection. Naringenin has hypocholesterolemic, antioxidant, free radical scavenger, anti-cancer, anti-inflammatory, neuroprotective, carbohydrate metabolism promoter, and immune system modulator properties. Naringenin possesses potent antidepressant-like property via the central serotonergic and noradrenergic systems.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Int J Mol Sci.2024, 25(19):10660.
J Nat Med.2020, 74(1):65-75
Nutrients.2024, 16(16):2612.
Biomolecules.2023, 13(2):227.
Processes 2021, 9(5),894.
In Vivo.2022, 36(3):1136-1143.
Mol Biol Rep.2024, 51(1):117.
J Pain Res.2022, 15:3469-3478.
Curr Issues Mol Biol.2022, 44(10):5106-5116.
Phytomedicine.2022, 102:154183.
Related and Featured Products
Int J Mol Med. 2012 Jul;30(1):204-10.
Naringenin attenuates the release of pro-inflammatory mediators from lipopolysaccharide-stimulated BV2 microglia by inactivating nuclear factor-κB and inhibiting mitogen-activated protein kinases.[Pubmed:
22552813 ]
Naringenin, one of the most abundant flavonoids in citrus fruits and grapefruits, has been reported to exhibit anti-inflammatory and antitumor activities. However, the cellular and molecular mechanisms underlying the Naringenin anti-inflammatory activity are poorly understood.
METHODS AND RESULTS:
In this study, we conducted an investigation of the inhibitory effects of Naringenin on the production of lipopolysaccharide (LPS)-induced pro-inflammatory mediators in BV2 microglial cells. We found that pre-treatment with Naringenin prior to treatment with LPS significantly inhibited excessive production of nitric oxide (NO) and prostaglandin E2 (PGE2) in a dose-dependent manner. The inhibition was associated with downregulation of inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2) expression. Naringenin also attenuated the production of pro-inflammatory cytokines and chemokines, including interleukin-1β (IL-1β), tumor necrosis factor-α (TNF-α) and monocyte chemoattractant protein-1 (MCP-1) by suppressing expression of mRNAs for these proteins. In addition, the molecular mechanism underlying Naringenin-mediated attenuation in BV2 cells has a close relationship to suppressing translocation of the nuclear factor-κB (NF-κB) p65 subunit into the nucleus and the phosphorylation of Akt and mitogen-activated protein kinases (MAPKs).
CONCLUSIONS:
These findings suggest that Naringenin may provide neuroprotection through suppression of pro-inflammatory pathways in activated BV2 microglial cells.
J Lipid Res. 2001 May;42(5):725-34.
Secretion of hepatocyte apoB is inhibited by the flavonoids, naringenin and hesperetin, via reduced activity and expression of ACAT2 and MTP.[Pubmed:
11352979]
The citrus flavonoids, Naringenin and hesperetin, lower plasma cholesterol in vivo. However, the underlying mechanisms are not fully understood.
METHODS AND RESULTS:
The ability of these flavonoids to modulate apolipoprotein B (apoB) secretion and cellular cholesterol homeostasis was determined in the human hepatoma cell line, HepG2. apoB accumulation in the media decreased in a dose-dependent manner following 24-h incubations with Naringenin (up to 82%, P < 0.00001) or hesperetin (up to 74%, P < 0.002). Decreased apoB secretion was associated with reduced cellular cholesteryl ester mass. Cholesterol esterification was decreased, dose-dependently, up to 84% (P < 0.0001) at flavonoid concentrations of 200 microM. Neither flavonoid demonstrated selective inhibition of either form of acyl CoA:cholesterol acyltransferase (ACAT) as determined using CHO cells stably transfected with either ACAT1 or ACAT2. However, in HepG2 cells, ACAT2 mRNA was selectively decreased (- 50%, P < 0.001) by both flavonoids, whereas ACAT1 mRNA was unaffected. In addition, Naringenin and hesperetin decreased both the activity (- 20% to - 40%, P < 0.00004) and expression (- 30% to - 40%, P < 0.02) of microsomal triglyceride transfer protein (MTP). Both flavonoids caused a 5- to 7-fold increase (P < 0.02) in low density lipoprotein (LDL) receptor mRNA, which resulted in a 1.5- to 2-fold increase in uptake and degradation of (125)I-LDL.
CONCLUSIONS:
We conclude that both Naringenin and hesperetin decrease the availability of lipids for assembly of apoB-containing lipoproteins, an effect mediated by 1) reduced activities of ACAT1 and ACAT2, 2) a selective decrease in ACAT2 expression, and 3) reduced MTP activity. Together with an enhanced expression of the LDL receptor, these mechanisms may explain the hypocholesterolemic properties of the citrus flavonoids.
Life Sci. 2005 Mar 18;76(18):2125-35.
Naringenin attenuates cisplatin nephrotoxicity in rats.[Pubmed:
15826879 ]
METHODS AND RESULTS:
The effect of Naringenin (NAR), a naturally occurring citrus flavanone, on the acute nephrotoxicity produced by cisplatin (7 mg/kg, i.v.) was investigated in the rat. Oral administration of NAR (20 mg/kg/day) for 10 days, starting 5 days before cisplatin single i.v. injection, produced significant protection of renal function. NAR reduced the extent of cisplatin-induced nephrotoxicity, as evidenced by significant reduction in serum urea and creatinine concentrations, decreased polyuria, reduction in body weight loss, marked reduction in urinary fractional sodium excretion and glutathione S-transferase (GST) activity, and increased creatinine clearance. Cisplatin-induced alterations in renal cortex lipid peroxides and GST activity were markedly improved by NAR. Cisplatin-induced alterations in renal cortex antioxidant defense system were greatly prevented by NAR. In cisplatin-NAR combined treatment group, antioxidant enzymes namely superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), and catalase (CAT) were significantly increased to 54.5, 30.3 and 35.6%, respectively compared to cisplatin treated group. Platinum renal content was not affected by NAR treatment.
CONCLUSIONS:
The results provide further insight into the mechanisms of cisplatin-induced nephrotoxicity and confirm the antioxidant potential of NAR.
Exp Toxicol Pathol. 2010 Mar;62(2):171-81.
Cadmium-induced hepatotoxicity in rats and the protective effect of naringenin.[Pubmed:
19409769]
This experiment pertains to the protective role of Naringenin against cadmium (Cd)-induced oxidative stress in the liver of rats. Cadmium is a major environmental pollutant and is known for its wide toxic manifestations. Naringenin is a naturally occurring citrus flavonone which has been reported to have a wide range of pharmacological properties.
METHODS AND RESULTS:
In the present investigation cadmium (5mg/kg) was administered orally for 4 weeks to induce hepatotoxicity. Liver damage induced by cadmium was clearly shown by the increased activities of serum hepatic marker enzymes namely aspartate transaminase (AST), alanine transaminase (ALT), alkaline phosphatase (ALP), lactate dehydrogenase (LDH), gamma glutamyl transferase (GGT) and serum total bilirubin (TB) along with the increased level of lipid peroxidation indices (thiobarbituric acid reactive substances (TBARS) and lipid hydroperoxides) and protein carbonyl contents in liver. The toxic effect of cadmium was also indicated by significantly decreased levels of enzymatic antioxidants (superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx) and glutathione S-transferase (GST)) and non-enzymatic antioxidants (reduced glutathione (GSH), vitamin C and vitamin E). Administration of Naringenin at a dose of (50mg/kg) significantly reversed the activities of serum hepatic marker enzymes to their near-normal levels when compared to Cd-treated rats. In addition, Naringenin significantly reduced lipid peroxidation and restored the levels of antioxidant defense in the liver. The histopathological studies in the liver of rats also showed that Naringenin (50mg/kg) markedly reduced the toxicity of cadmium and preserved the normal histological architecture of the tissue.
CONCLUSIONS:
The present study suggested that Naringenin may be beneficial in ameliorating the cadmium-induced oxidative damage in the liver of rats.
Free Radic Res. 2005 Oct;39(10):1119-25.
Neuroprotective properties of the natural phenolic antioxidants curcumin and naringenin but not quercetin and fisetin in a 6-OHDA model of Parkinson's disease.[Pubmed:
16298737 ]
Although the cause of dopaminergic cell death in Parkinson's disease (PD) remains unknown, oxidative stress has been strongly implicated. Because of their ability to combat oxidative stress, diet derived phenolic compounds continue to be considered as potential agents for long-term use in PD.
This study was aimed at investigating whether the natural phenolic compounds curcumin, Naringenin, quercetin, fisetin can be neuroprotective in the 6-OHDA model of PD.
METHODS AND RESULTS:
Unilateral infusion of 6-OHDA into the medial forebrain bundle produced a significant loss of tyrosine hydroxylase (TH)-positive cells in the substantia nigra (SN) as well as a decreased of dopamine (DA) content in the striata in the vehicle-treated animals. Rats pretreated with curcumin or Naringenin showed a clear protection of the number of TH-positive cells in the SN and DA levels in the striata. However, neither pretreatment with quercetin nor fisetin had any effects on TH-positive cells or DA levels.
CONCLUSIONS:
The ability of curcumin and Naringenin to exhibit neuroprotection in the 6-OHDA model of PD may be related to their antioxidant capabilities and their capability to penetrate into the brain.
Prog Neuropsychopharmacol Biol Psychiatry. 2010 Oct 1;34(7):1223-8.
Involvement of monoaminergic system in the antidepressant-like effect of the flavonoid naringenin in mice.[Pubmed:
20603175 ]
Dietary flavonoids possess a multiplicity of neuroprotective actions in various central nervous pathophysiological conditions including depression.
METHODS AND RESULTS:
In this study, the neuropharmacological mechanism of the dietary flavonoid Naringenin was investigated in the mouse behavioral models of depression. For this purpose, we investigated the influence of pretreatment with the inhibitors of serotonin or noradrenaline synthesis, p-chlorophenylalanine methyl ester or α-methyl-p-tyrosine, respectively in the anti-immobility effect of Naringenin. Compared to the control group, Naringenin significantly decreased the immobility time after acute treatment in the mouse tail suspension test (10, 20 and 50 mg/kg), but not in the forced swimming test, without producing locomotor alteration in the open-field test. In addition, pretreatment of mice with p-chlorophenylalanine methyl ester (100 mg/kg) or α-methyl-p-tyrosine (100 mg/kg) prevented the anti-immobility effect of Naringenin (20 mg/kg) in the tail suspension test.
CONCLUSIONS:
Taken together, this data demonstrated that Naringenin possessed potent antidepressant-like property via the central serotonergic and noradrenergic systems. Thus, our findings suggest the therapeutic potential of this dietary flavonoid in central nervous system disorders especially depression where monoaminergic systems are involved.
Biochem Pharmacol. 1995 Oct 26;50(9):1485-93.
Naringenin: a weakly estrogenic bioflavonoid that exhibits antiestrogenic activity.[Pubmed:
7503800]
METHODS AND RESULTS:
Treatment of immature 21-day-old female Sprague-Dawley rats with 17 beta-estradiol (E2) (0.5 microgram/rat) caused a significant increase in uterine wet weight, DNA synthesis, progesterone receptor (PR) binding, and peroxidase activity.
At doses as high as 40 mg/rat, the bioflavonoid Naringenin did not cause a significant increase in any of these E2-induced responses. However, in rats cotreated with E2 (0.5 microgram/rat) plus Naringenin (30 mg/rat); there was a significant decrease in E2-induced uterine wet weight, DNA synthesis, PR binding, and peroxidase activity, indicating that Naringenin exhibits antiestrogenic activity in the immature rodent uterus. The binding of uterine nuclear extracts to a 32P-labeled estrogen responsive element (ERE) or progesterone responsive element (PRE) was determined using gel electrophoretic band shift assays. Incubation of [32P]ERE with uterine nuclear extracts from rats treated with Naringenin or E2 resulted in the formation of estrogen receptor (ER):ERE complexes; a higher mobility complex was prominent in the extracts from E2-treated rats, whereas a lower mobility complex was observed using nuclear extracts from Naringenin-treated animals. There was a significant decrease in the intensity of the E2-induced complex using nuclear extracts from rats treated with E2 plus Naringenin. In contrast, transformed cytosol from control rats gave an intense ER:ERE complex, whereas the intensity of the band was decreased markedly using transformed uterine cytosol from treated rats. Formation of a PR:PRE complex was also determined using transformed uterine cytosol. Cytosol from E2-treated rats gave an intense retarded band, whereas only weak bands were observed using cytosols from DMSO- (solvent), Naringenin-, or Naringenin plus E2-treated cells. The results of in vitro studies showed that 1 nM E2 increased (3- to 4-fold) the growth of MCF-7 human breast cancer cells, whereas 1-1000 nM Naringenin had no effect on cell proliferation. In cells cotreated with 1 nM E2 plus 1000 nM Naringenin, there was a significant decrease in E2-induced cell growth. In MCF-7 cells transiently transfected with a pS2 promoter-regulated luciferase reporter gene, Naringenin exhibited weak estrogenic activity. In cells cotreated with 0.1 or 1.0 microM Naringenin plus 1 nM E2, Naringenin inhibited E2-induced luciferase activity.
CONCLUSIONS:
The results of these studies confirmed that Naringenin is a weak estrogen that also exhibits partial antiestrogenic activity in the female rat uterus and MCF-7 human breast cancer cells.
Biochem Biophys Res Commun. 2001 Jun 15;284(3):681-8.
Anti-atherogenic effect of citrus flavonoids, naringin and naringenin, associated with hepatic ACAT and aortic VCAM-1 and MCP-1 in high cholesterol-fed rabbits.[Pubmed:
11396955]
The anti-atherogenic effects of the citrus flavonoids, naringin and Naringenin, were evaluated in high cholesterol-fed rabbits.
METHODS AND RESULTS:
At 3 months of age, 30 male New Zealand White (NZW) rabbits were divided into three groups (n = 10 per group). The rabbits were fed a 1% cholesterol diet alone (control group) or a diet supplemented with either 0.1% naringin or 0.05% Naringenin for 8 weeks. The plasma lipoprotein levels, total cholesterol, triglyceride, and high-density lipoprotein showed no significant differences in the control and experimental groups. Hepatic acyl-CoA:cholesterol acyltransferase (ACAT) activity was slightly low in naringin (5.0%)- and Naringenin (15.0%)-fed rabbits, compared to control group. The aortic fatty streak areas were significantly lower in both the naringin (19.2 +/- 5.6%)- and Naringenin (18.1 +/- 6.5%)-supplemented groups than in the control group (60.4 +/- 14.0%). The expression levels of vascular cell adhesion molecule-1 (VCAM-1) and monocyte chemotactic protein-1 (MCP-1), by semiquantitative RT-PCR analysis of the thoracic aorta, were significantly lower in the flavonoids supplemented groups than in the control group.
CONCLUSIONS:
These results suggest that the anti-atherogenic effect of the citrus flavonoids, naringin and Naringenin, is involved with a decreased hepatic ACAT activity and with the downregulation of VCAM-1 and MCP-1 gene expression.
Other References Information
2017 Feb 3;7:41864.
The citrus flavanone naringenin impairs dengue virus replication in human cells[Pubmed:
28157234]
Dengue is one of the most significant health problems in tropical and sub-tropical regions throughout the world. Nearly 390 million cases are reported each year. Although a vaccine was recently approved in certain countries, an anti-dengue virus drug is still needed. Fruits and vegetables may be sources of compounds with medicinal properties, such as flavonoids. This study demonstrates the anti-dengue virus activity of the citrus flavanone Naringenin, a class of flavonoid. Naringenin prevented infection with four dengue virus serotypes in Huh7.5 cells. Additionally, experiments employing subgenomic RepDV-1 and RepDV-3 replicon systems confirmed the ability of Naringenin to inhibit dengue virus replication. Antiviral activity was observed even when Naringenin was used to treat Huh7.5 cells 24 h after dengue virus exposure. Finally, Naringenin anti-dengue virus activity was demonstrated in primary human monocytes infected with dengue virus sertoype-4, supporting the potential use of Naringenin to control dengue virus replication. In conclusion, Naringenin is a suitable candidate molecule for the development of specific dengue virus treatments.