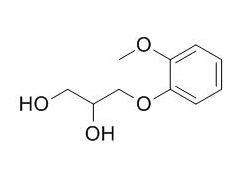
Guaifenesin
Guaifenesin is also a centrally acting skeletal muscle relaxant. Guaifenesin has antitussive effects, it can inhibit the cough-reflex sensitivity in acute viral cough, it is an expectorant commonly by thinning the mucus or phlegm in the lungs.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Applied Biological Chem. 2020, 26(63).
Environ Toxicol.2024, tox.24246
J Agric Food Chem.2016, 64(35):6783-90
Pharmaceuticals (Basel).2020, 13(10):302.
Horticulture Research2023, uhad259
BMC Complement Med Ther. 2020, 20(1):91.
Food Res Int.2020, 128:108778
Plants (Basel).2020, 9(11):1555.
Am J Chin Med.2023, 51(7):1675-1709.
Agronomy2020, 10(3),388.
Related and Featured Products
J Clin Pharmacol. 2015 Dec 3. doi: 10.1002/jcph.682.
Guaifenesin Pharmacokinetics Following Single-Dose Oral Administration in Children Aged 2 to 17 Years.[Pubmed:
26632082]
METHODS AND RESULTS:
This study characterized Guaifenesin pharmacokinetics in children aged 2 to 17 years (n = 40) who received a single oral dose of Guaifenesin (age-based doses of 100-400 mg) 2 hours after breakfast. Plasma samples were obtained before and for 8 hours after dosing and analyzed for Guaifenesin using liquid chromatography-tandem mass spectrometry. Pharmacokinetic parameters were estimated using noncompartmental methods, relationships with age were assessed using linear regression, and dose proportionality was assessed on 95% confidence intervals. Based on the upper dose recommended in the monograph (for both children and adolescents), area under the curve from time zero to infinity and maximum plasma concentration both increased with age. However, when comparing the upper dose for children aged 2 to 11 years with the lower dose for adolescents aged 12 to 17 years, similar systemic exposure was observed. As expected due to increasing body size, oral clearance (CLo ) and terminal volume of distribution (Vz /F) increased with age. Due to a larger increase in Vz /F than CLo , an increase in terminal exponential half-life was also observed.
CONCLUSIONS:
Allometric scaling indicated no maturation-related changes in CLo and Vz /F.
J Pain Res. 2017 Mar 21;10:669-678.
Efficacy and safety of guaifenesin for upper back, neck, and shoulder pain: a Phase II proof-of-concept, multicenter, placebo-controlled, repeat-dose, parallel-group study.[Pubmed:
28356767]
Guaifenesin, an over-the-counter (OTC) expectorant, has exhibited muscle relaxant effects preclinically and clinically. This proof-of-principle study explored whether OTC doses of Guaifenesin can provide relief from acute upper back, neck, or shoulder muscle spasm and pain.
METHODS AND RESULTS:
This multicenter, placebo-controlled, repeat-dose, parallel study randomly assigned adults experiencing acute pain and muscle spasm in their upper back, neck, or shoulder to Guaifenesin 600 or 1200 mg or matched placebo twice daily (BID) in a 2:2:1:1 ratio for 7 days. The primary end point was the change from baseline in muscle spasm relief, measured using an 11-point numeric rating scale (0=not present to 10=unbearable) recorded twice daily and averaged over the 7-day treatment period. Analyses were performed using a linear mixed model that included treatment as a fixed effect and site as a random effect.
A total of 77 subjects were included in the 4 treatment groups. Least squares mean muscle spasm score over 7 days was 1.77 with Guaifenesin 1200 mg, 1.42 with its matched placebo, 1.53 with Guaifenesin 600 mg, and 1.74 with its matched placebo. Treatment with Guaifenesin 1200 mg BID provided 25% greater reduction in mean muscle spasm over its matched placebo and 16% greater reduction than Guaifenesin 600 mg BID. These differences were not statistically significant. Based on comparisons of absolute mean values, a consistent directional change in effect was observed, suggesting some benefit from placebo to lower-to-upper doses of Guaifenesin with regard to muscle spasm, tension, pain, discomfort, and relaxation. No severe or serious adverse events were reported.
CONCLUSIONS:
Results suggest the potential for OTC dose of Guaifenesin 1200 mg BID to provide symptomatic relief of upper back musculoskeletal pain and spasm. Confirmation of this preliminary result in a larger, adequately powered study is needed.
Respir Med. 2009 Jun;103(6):902-6.
Inhibition of cough-reflex sensitivity by benzonatate and guaifenesin in acute viral cough.[Pubmed:
19121573]
Acute cough due to viral upper respiratory tract infection (URI) is the most common form of cough and accounts for tremendous expenditure on prescription and non-prescription cough products worldwide. However, few agents have been shown in properly conducted clinical trials to be effective for cough due to URI.
METHODS AND RESULTS:
The present study evaluated the effect of benzonatate 200mg (B), Guaifenesin 600 mg (G), their combination (B+G), and placebo (P) on capsaicin-induced cough in 30 adult nonsmokers with acute URI. On 3 separate days within a 7-day period, 1h after ingesting randomly assigned study drug in a double-blind fashion, subjects underwent capsaicin cough challenge testing, which involved inhalation of incremental doubling concentrations of capsaicin until the concentration of capsaicin inducing 5 or more coughs (C(5)) was attained. Each subject received 3 of 4 possible study drugs. G (p=0.01) but not B (p=NS) inhibited cough-reflex sensitivity (log C(5)) relative to P. The combination of B+G suppressed capsaicin-induced cough to a greater degree than B alone (p<0.001) or G alone (p=0.008). The mechanism by which the combination of B+G causes a potentiation of antitussive effect remains to be elucidated.
CONCLUSIONS:
Our results suggest that B+G may be an effective therapy for acute cough due to the common cold (URI).
J Vet Pharmacol Ther. 2016 Jan 14. doi: 10.1111/jvp.12287.
Pharmacokinetics of guaifenesin following administration of multiple doses to exercised Thoroughbred horses.[Pubmed:
26763117]
Guaifenesin is an expectorant commonly used in performance horses to aid in the clearance of mucus from the airways. Guaifenesin is also a centrally acting skeletal muscle relaxant and as such is a prohibited drug with withdrawal necessary prior to competition. To the authors' knowledge, there are no reports in the literature describing single or multiple oral administrations of Guaifenesin in the horse to determine a regulatory threshold and related withdrawal time.
METHODS AND RESULTS:
Therefore, the objective of the current study was to describe the pharmacokinetics of Guaifenesin following oral administration in order to provide data upon which appropriate regulatory recommendations can be established. Nine exercised Thoroughbred horses were administered 2 g of Guaifenesin orally BID for a total of five doses. Blood samples were collected immediately prior to drug administration and at various times postadministration. Serum Guaifenesin concentrations were determined and pharmacokinetic parameters calculated. Guaifenesin was rapidly absorbed (Tmax of 15 min) following oral administration. The Cmax was 681.3 ± 323.8 ng/mL and 1080 ± 732.8 following the first and last dose, respectively. The serum elimination half-life was 2.62 ± 1.24 h. Average serum Guaifenesin concentrations remained above the LOQ of the assay (0.5 ng/mL) by 48 h postadministration of the final dose in 3 of 9 horses.