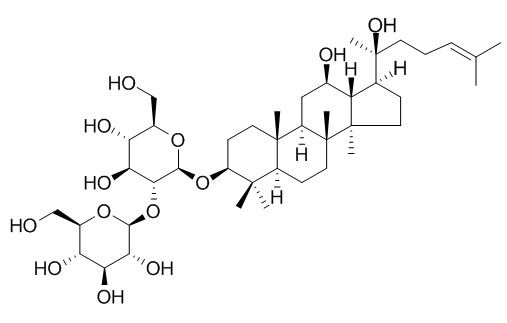
Ginsenoside Rg3
Ginsenoside Rg3 is the main component of Red ginseng, it inhibits Na+ and hKv1.4 channel with IC50s of 32.2±4.5 and 32.6±2.2 μM, respectively, it also inhibits Aβ levels, NF-κB activity, and COX-2 expression. Ginsenoside-Rg3 possesses an ability to inhibit the lung metastasis of tumor cells, and the mechanism of its antimetastatic effect is related to inhibition of the adhesion and invasion of tumor cells, and also to anti-angiogenesis activity. It is a novel drug, capable of inhibiting the early of scarring (HS) and later HS.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Eur Endod J.2020, 5(1):23-27.
Antioxidants (Basel).2024, 13(3):340.
Enzyme and Microbial Technology2022, 110002.
Food Chem.2019, 278:683-691
Prev Nutr Food Sci.2025, 30(1):92-100.
LWT2020, 126:109313
Processes 2021, 9(5),894.
Planta Medica International2022, 9(01):e108-e115.
Int J Mol Sci.2023, 24(24):17589.
Preprints2022, 202211.0388.v1.
Related and Featured Products
Biol Pharm Bull. 2015;38(1):102-8.
Stereoisomer-specific anticancer activities of ginsenoside Rg3 and Rh2 in HepG2 cells: disparity in cytotoxicity and autophagy-inducing effects due to 20(S)-epimers.[Pubmed:
25744465]
Autophagy has been an emerging field in the treatment of hepatic carcinoma since anticancer therapies were shown to ignite autophagy in vitro and in vivo. Here we report that Ginsenoside Rg3 and Rh2, major components of red ginseng, induce apoptotic cell death in a stereoisomer-specific fashion.
METHODS AND RESULTS:
The 20(S)-forms of Ginsenoside Rg3 and Rh2, but not their respective 20(R)-forms, promoted cell death in a dose-dependent manner accompanied by downregulation of Bcl2 and upregulation of Fas, resulting in apoptosis of HepG2 cells with poly ADP ribose polymerase cleavage. The LD50 value [45 μM for Rg3(S), less than 10 μM for Rh2(S)] and gross morphological electron microscopic observation revealed more severe cellular damage in cells treated with Rh2(S) than in those treated with Rg3(S). Both Rg3(S) and Rh2(S) also induced autophagy when undergoing induced apoptosis. Inhibition of autophagy with lysosomotrophic agents significantly potentiated the cellular damage, implying a favorable switch of the cell fate to tumor cell death. Blocking intracellular calcium with 1,2-bis(2-aminophenoxy)ethane-N,N,N',N'-tetraacetic acid tetrakis(acetoxymethyl ester) (BAPTA-AM) restored the cell death induced by both Rg3(S) and Rh2(S).
CONCLUSIONS:
Our results suggest that the 20(S)-forms of Ginsenoside Rg3 and Rh2 in red ginseng possess more potent antitumor activity with autophagy than their 20(R)-forms via calcium-dependent apoptosis.
Mol Med Rep. 2015 Jul;12(1):609-14.
Ginsenoside Rg3 sensitizes human non‑small cell lung cancer cells to γ-radiation by targeting the nuclear factor-κB pathway.[Pubmed:
25738799]
At present, it is elusive how non-small cell lung cancer (NSCLC) develops resistance to γ-radiation; however, the transcription factor nuclear factor-κB (NF-κB) and NF-κB-regulated gene products have been proposed as mediators. Ginsenoside Rg3 is a steroidal saponin, which was isolated from Panax ginseng. Ginsenoside Rg3 possesses high pharmacological activity and has previously been shown to suppress NF-κB activation in various types of tumor cell. Therefore, the present study aimed to determine whether Rg3 could suppress NF-κB activation in NSCLC cells and sensitize NSCLC to γ-radiation, using an NSCLC cell line and NSCLC xenograft.
METHODS AND RESULTS:
A clone formation assay and lung tumor xenograft experiment were used to assess the radiosensitizing effects of Ginsenoside Rg3. NF-κB/inhibitor of NF-κB (IκB) modulation was ascertained using an electrophoretic mobility shift assay and western blot analysis. NF-κB-regulated gene products were monitored by western blot analysis. The present study demonstrated that Ginsenoside Rg3 was able to sensitize A549 and H1299 lung carcinoma cells to γ-radiation and significantly enhance the efficacy of radiation therapy in C57BL/6 mice bearing a Lewis lung carcinoma cell xenograft tumor. Furthermore, Ginsenoside Rg3 suppressed NF-κB activation, phosphorylation of IκB protein and expression of NF-κB-regulated gene products (cyclin D1, c-myc, B-cell lymphoma 2, cyclooxygenase-2, matrix metalloproteinase-9 and vascular endothelial growth factor), a number of which were induced by radiation therapy and mediate radioresistance.
CONCLUSIONS:
In conclusion, the results of the present study suggested that Ginsenoside Rg3 may potentiate the antitumor effects of radiation therapy in NSCLC by suppressing NF-κB activity and NF-κB-regulated gene products, leading to the inhibition of tumor progression.
J Immunol Res . 2016;2016:7521601.
Anti-Inflammatory Effects of Ginsenoside Rg3 via NF- κ B Pathway in A549 Cells and Human Asthmatic Lung Tissue[Pubmed:
28116321]
Objective. There is limited information of the anti-inflammatory effects of Rg3 on inflamed lung cells and tissues. Therefore, we confirmed the anti-inflammatory mechanism of Ginsenoside Rg3 in inflamed human airway epithelial cells (A549) and tissues whether Rg3 regulates nuclear factor kappa B (NF-κB) activity. Methods. To induce the inflammation, IL-1β (10 ng/ml) was treated to A549 cells for 4 h. The effects of Rg3 on NF-κB activity and COX-2 expression were evaluated by western blotting analysis in both IL-1β-induced inflamed A549 cell and human asthmatic airway epithelial tissues. Using multiplex cytokines assay, the secretion levels of NF-κB-mediated cytokines/chemokines were measured. Result. Rg3 showed the significant inhibition of NF-κB activity thereby reduced COX-2 expression was determined in both IL-1β-induced inflamed A549 cell and human asthmatic airway epithelial tissues. In addition, among NF-κB-mediated cytokines, the secretion levels of IL-4, TNF-α, and eotaxin were significantly decreased by Rg3 in asthma tissues. Even though there was no significant difference, IL-6, IL-9, and IL-13 secretion showed a lower tendency compared to saline-treated human asthmatic airway epithelial tissues. Conclusion. The results from this study demonstrate the potential of Rg3 as an anti-inflammatory agent through regulating NF-κB activity and reducing the secretion of NF-κB-mediated cytokines/chemokines.
Chem Biol Interact. 2015 May 25;233:25-34.
Novel roles of ginsenoside Rg3 in apoptosis through downregulation of epidermal growth factor receptor.[Pubmed:
25824408]
Ginsenoside Rg3 (Rg3), a pharmacologically active compound from red ginseng, has been reported to induce cell death in various cancer cell lines, although the specific mechanisms have not been well established. In the present study, Ginsenoside Rg3 treatment to A549 human lung adenocarcinoma led to cell death via not only apoptotic pathways but also the downregulation of epidermal growth factor receptor (EGFR).
METHODS AND RESULTS:
We used cross-linker and cell enzyme-linked immunosorbent assays to show that Ginsenoside Rg3 inhibited EGFR dimerization by EGF stimulation and caused EGFR internalization from the cell membrane. Among several important phosphorylation sites in cytoplasmic EGFR, Ginsenoside Rg3 increased the phosphorylation of tyrosine 1045 (pY1045) and serine 1046/1047 (pS1046/1047) for EGFR degradation and coincidently, attenuated pY1173 and pY1068 for mitogen-activated protein kinase activity. These effects were amplified under EGF-pretreated Ginsenoside Rg3 stimulation. In vivo experiments showed that the average volume of the tumors treated with 30mg/kg of Ginsenoside Rg3 was significantly decreased by 40% compared with the control. Through immunohistochemistry, we detected the fragmentation of DNA, the accumulation of Ginsenoside Rg3, and the reduction of EGFR expression in the Ginsenoside Rg3-treated groups.
CONCLUSIONS:
Here, we provide the first description of the roles of Ginsenoside Rg3 in the reduction of cell surface EGFR, the attenuation of EGFR signal transduction, and the eventual activation of apoptosis in A549 human lung adenocarcinoma.
Acta Biomater. 2013 Dec;9(12):9461-73.
In vivo inhibition of hypertrophic scars by implantable ginsenoside-Rg3-loaded electrospun fibrous membranes.[Pubmed:
23938200]
Clinically, hypertrophic scarring (HS) is a major concern for patients and has been a challenge for surgeons, as there is a lack of treatments that can intervene early in the formation of HS.
This study reports on a Chinese drug, 20(R)-Ginsenoside Rg3 (GS-Rg3), which can inhibit in vivo the early formation of HS and later HS hyperplasia by inducing the apoptosis of fibroblasts, inhibiting inflammation and down-regulating VEGF expression.
METHODS AND RESULTS:
Implantable biodegradable GS-Rg3-loaded poly(l-lactide) (PLA) fibrous membranes were successfully fabricated using co-electrospinning technology to control drug release and improve drug utilization. The in vivo releasing time of GS-Rg3 lasts for 3 months, and the drug concentration released in rabbits can be controlled by varying the drug content of the electrospun fibers. Histological observations of HE staining indicate that GS-Rg3/PLA significantly inhibits the HS formation, with obvious improvements in terms of dermis layer thickness, epidermis layer thickness and fibroblast proliferation. The results of immunohistochemistry staining and Masson's trichrome staining demonstrate that GS-Rg3/PLA electrospun fibrous membranes significantly inhibit HS formation, with decreased expression of collagen fibers and microvessels. VEGF protein levels are much lower in the group treated with GS-Rg3/PLA eletrospun membranes compared with other groups.
CONCLUSIONS:
These results demonstrate that GS-Rg3 is a novel drug, capable of inhibiting the early formation of HS and later HS hyperplasia. GS-Rg3/PLA electrospun membrane is a very promising new treatment for early and long-term treatment of HS.
Mol Med Rep. 2015 Jun;11(6):4518-24.
Ginsenoside Rg3 attenuates myocardial ischemia/reperfusion injury via Akt/endothelial nitric oxide synthase signaling and the B‑cell lymphoma/B‑cell lymphoma‑associated X protein pathway.[Pubmed:
25672441]
Previous studies have suggested that Ginsenoside Rg3 (GSRg3) extract from the medicinal plant Panax ginseng, may increase nitric oxide production via increases in the phosphorylation and expression of endothelial nitric oxide synthase (eNOS).
METHODS AND RESULTS:
The present study used an in vitro neonatal rat cardiomyocyte (NRC) model of anoxia‑reoxygenation injury and an in vivo rat model of myocardial ischemia/reperfusion (MI/R) injury. Hemodynamic, histopathological and biochemical assessment of the myocardial injury was performed and the expression levels of lactate dehydrogenase (LDH), superoxide dismutase and creatine kinase (CK) were measured in serum from the animal model, which may reflect myocardial injury. NRC injury was determined using a Cell Counting kit‑8. The GSRg3 anti‑apoptotic effects were assessed using flow cytometry to investigate the number of early‑late apoptotic cells and western blot analysis was performed to analyze the protein expression levels of caspase‑3, caspase‑9, B‑cell lymphoma‑2 (Bcl‑2), phosphorylated (p‑)Akt and eNOS. The results suggested that pretreatment with GSRg3 (60 mg/kg) significantly improved rat cardiac function, as demonstrated by increased left ventricular systolic pressure, heart rate and first derivative of left ventricular pressure. GSRg3 also reduced the size of the myocardial infarct and LDH/CK levels in the blood following MI/R. In vitro investigations revealed that GSRg3 (10 mM) decreased NRC apoptosis through inhibiting the activation of caspase‑3 and caspase‑9, and increasing the expression levels of p‑Akt, eNOS and the ratio of Bcl‑2/Bcl‑2‑associated X protein (Bax).
CONCLUSIONS:
Overall, the present study revealed that GSRg3 mediated a cardioprotective effect against MI/R‑induced apoptosis via Akt/eNOS signaling and the Bcl‑2/Bax pathway.
J Nat Med. 2012 Jul;66(3):476-85.
Red ginseng and 20(S)-Rg3 control testosterone-induced prostate hyperplasia by deregulating androgen receptor signaling.[Pubmed:
22101440 ]
Since prostate growth is governed by the androgen signaling pathway, blockade of the pathway is regarded as an appropriate strategy for the treatment of benign prostatic hyperplasia (BPH). Panax ginseng is known to have various pharmacological activities. Of several products of its root, red ginseng, having many bioactive ginsenosides, is most popularly used in Korea, and recently has been reported to control the proliferation of cancer cells. We here tested the effect of a water extract of Korean red ginseng (WKRG) on testosterone-induced prostate hyperplasia.
METHODS AND RESULTS:
WKRG (daily intraperitoneal injection) prevented prostate overgrowth and epithelial thickening induced by testosterone in rats, and suppressed a rat prostate kallikrein-S3. In human prostate cells, WKRG inhibited testosterone-induced cell proliferation, arrested cell cycle by inducing p21 and p27, and induced apoptosis. Testosterone-induced expression of human kallikrein-3 mRNA and activation of androgen receptor (AR) were effectively inhibited by WKRG. Of the major ginsenosides included in WKRG, 20(S)-Rg3 was identified to repress AR activity and to attenuate prostate cell growth during testosterone stimulation. Moreover, 20(S)-Rg3 downregulated AR by facilitating the degradation of AR protein. WKRG and 20(S)-Rg3 were found to have new pharmacological activities against testosterone-induced prostate overgrowth.
CONCLUSIONS:
Given that red ginseng has been used safely in Asia for 1000 years, red ginseng and 20(S)-Rg3 could be potential therapeutic regimens for treating BPH.