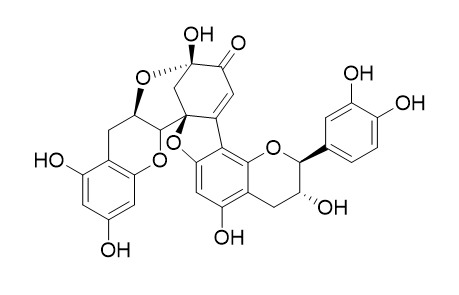
Dehydrodicatechin A
The peroxidase-mediated (+)-catechin oxidation was differentially inhibited by dehydrodicatechin A, showing a competitive type inhibition and a k(I) of 6.4 microM. Dehydrobisphenol A has anticancer and antioxidant activity.
Inquire / Order:
manager@chemfaces.com
Technical Inquiries:
service@chemfaces.com
Tel:
+86-27-84237783
Fax:
+86-27-84254680
Address:
1 Building, No. 83, CheCheng Rd., Wuhan Economic and Technological Development Zone, Wuhan, Hubei 430056, PRC
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to
24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
Food Res Int.2020, 128:108778
Food Chem.2022, 373(Pt B):131364.
Evid Based Complement Alternat Med.2020, 2020:2584783.
Pharmacol Rep.2019, 71(2):289-298
Phytother Res.2019, 33(3):676-689
Industrial Crops and Products2020, 146:112186
J Pharmaceut Biomed2020, 178:112894
In Vitro Cellular & Developmental Biology - Plant 2021, 57:874�C882.
J Chromatogr A.2022, 1685:463640.
J AOAC Int.2024, qsae028.
Related and Featured Products
J Agric Food Chem. 2002 Feb 27;50(5):1218-1224.
Comparative study of the products of the peroxidase-catalyzed and the polyphenoloxidase-catalyzed (+)-catechin oxidation. Their possible implications in strawberry (Fragaria x ananassa) browning reactions[Pubmed:
11853507]
The peroxidase- and polyphenoloxidase-catalyzed oxidations of (+)-catechin yield several products showing different degrees of polymerization, which are apparently responsible for the pigment decay and the associated browning reaction that occurs in processed strawberry fruits and their derived foods. In this work, we have purified both peroxidase and polyphenoloxidase from Oso Grande cv. strawberry fruits, and comparatively analyzed the products of their enzyme-mediated (+)-catechin oxidation. The joint analysis by reversed-phase and size-exclusion HPLC of the (+)-catechin oxidation products obtained with both enzymes indicate that they were qualitatively the same: dehydrodicatechin B4, a (+)-catechin quinone methide, Dehydrodicatechin A, a (+)-catechin trimer, and a (+)-catechin oligomer with polymerization degree equal to or greater than 5. The main quantitative differences between the oxidative reactions were the great amount of oligomer formed in the case of the polyphenoloxidase-mediated reaction and the low amount of (+)-catechin reacted in the case of the peroxidase-mediated reaction. One of the possible reasons for such low levels of (+)-catechin consumption in the case of the peroxidase-mediated reaction was the possible inhibition by products of the enzyme-catalyzed oxidation. In fact, the peroxidase-mediated (+)-catechin oxidation was differentially inhibited by Dehydrodicatechin A, showing a competitive type inhibition and a k(I) of 6.4 microM. In light of these observations, these results suggest that brown polymer formation, estimated as oligomeric compounds resulting from (+)-catechin oxidation, in strawberries is mainly due to polyphenoloxidase, and although peroxidase also plays an important role, it is apparently auto-regulated by product (Dehydrodicatechin A) inhibition.
Z Naturforsch C J Biosci . Sep-Oct 2007;62(9-10):656-660.
Evaluation of cytotoxic compounds from calligonum comosum L. growing in Egypt[Pubmed:
18069236]
Calligonum comosum (Polygonaceae), an Egyptian desert plant, was extracted and fractionated using petroleum ether, methylene chloride, and ethyl acetate. The total methanolic extract and other fractions were tested for their anticancer activity using Ehrlich ascites, brine shrimp and antioxidant assays. Ethyl acetate fraction proved to be the most active in all assays. Eight compounds were isolated, purified, and identified from this fraction as (+)-catechin (1), Dehydrodicatechin A (2), kaempferol-3-O-rhamnopyranoside (3), quercitrin (quercetin-3-O-rhamnopyranoside) (4), beta-sitosterol-3-O-glucoside (5), isoquercitrin (quercetin-3-O-glucopyranoside) (6), kaempferol-3-O-glucuronide (7), and mequilianin (quercetin-3-O-glucuronide) (8). All isolated compounds were tested for their cytotoxicity and antioxidant activity. Compound 2 showed the best cytotoxic and antioxidant activity.
J Sep Sci . 2021 Dec;44(24):4422-4430.
Isolation and purification of flavonoids from Euonymus alatus by high-speed countercurrent chromatography and neuroprotective effect of rhamnazin-3-O-rutinoside in vitro[Pubmed:
34670011]
The flavonoids from Euonymus alatus exhibit many biological activities including significant antioxidant, anti-inflammatory, anti-cancer. In this work, a high-speed countercurrent chromatography method for the isolation and purification of flavonoids from crude extracts of Euonymus alatus was established. The effects of several solvent systems on the separation efficiency of target compounds in the extract of Euonymus alatus were studied. The solvent system composed of n-hexane-ethyl acetate-methanol-water at a volume ratio of (3:5:3:5, v/v) was chosen, in which the lower phase was used as the mobile phase at the rotation speed of 800 rpm and flow rate of 2.0 mL/min. The three flavonoids were obtained and identified as patuletin-3-O-rutinoside, rhamnazin-3-O-rutinoside, and Dehydrodicatechin A by mass spectroscopy and nuclear magnetic resonance, and the quantities of patuletin-3-O-rutinoside, rhamnazin-3-O-rutinoside, and Dehydrodicatechin A were 2.2, 9.7, and 1.8 mg, respectively. The results indicated that high-speed countercurrent chromatography was a simple and efficient method for the isolation and purification of flavonoids from the crude extracts of Euonymus alatus. The cellular antioxidant activity experimental result indicated that rhamnazin-3-O-rutinoside could alleviate H2 O2 -induced oxidative stress.
Pharm Biol . 2016 Oct;54(10):2119-2126.
Flavonoids of Calligonum polygonoides and their cytotoxicity[Pubmed:
26922854]
Context Calligonum polygonoides L. subsp. comosum L' Hér. (Polygonaceae), locally known as "arta", is a slow-growing small leafless desert shrub. Objective Isolation, structure elucidation and evaluation of cytotoxic activity of flavonoids from C. polygonoides aerial parts. Materials and methods Flavonoids in the hydroalcoholic extract of the of C. polygonoides were isolated and purified using column chromatography and preparative HPLC. The structures of the isolated flavonoids were elucidated on the basis of spectroscopic data including 2D NMR techniques. The cytotoxic activity of the isolated flavonoids (6.25, 25, 50 and 100 μg/mL) was evaluated against liver HepG2 and breast MCF-7 cancer cell lines using sulphorhodamine-B assay. Results A new flavonoid, kaempferol-3-O-β-D-(6″-n-butyl glucuronide) (1), and 13 known flavonoids, quercetin 3-O-β-D-(6″-n-butyl glucuronide) (2), kaempferol-3-O-β-D-(6″-methyl glucuronide) (3), quercetin-3-O-β-D-(6″-methyl glucuronide) (4), quercetin-3-O-glucuronide (5), kaempferol-3-O-glucuronide (6), quercetin-3-O-α-rhamnopyranoside (7), astragalin (8), quercetin-3-O-glucopyranoside (9), taxifolin (10), (+)-catechin (11), Dehydrodicatechin A (12), quercetin (13), and kaempferol (14), were isolated from the aerial parts of C. polygonoides. Quercetin showed significant cytotoxic activity against HepG2 and MCF-7 cell lines with IC50 values of 4.88 and 0.87 μg/mL, respectively. Structure-activity relationships were analyzed by comparing IC50 values of several pairs of flavonoids differing in one structural element. Discussion and conclusion The activity against breast cancer cell lines decreased by glycosylation at C-3. The presence of 2,3-double bond in ring C, carbonyl group at C-4 and 3',4'-dihydroxy substituents in ring B are essential structural requirements for the cytotoxic activity against breast cancer cells.
J Asian Nat Prod Res . 2012;14(10):966-972.
Chemical constituents of Bauhinia aurea[Pubmed:
23046468]
A new dihydroflavonol glycoside dimer 6,6-bisastilbin (1) and a new nitrile-containing metabolite (Z)-5α,6β-dihydroxy-4β-methoxy-2-cyclohexene-Δ(1,α)-acetonitrile (2), together with three known analogs, bauhinin, bauhinilide, and Dehydrodicatechin A, have been isolated from an ethanol extract of Bauhinia aurea. Their structures were determined by spectroscopic and chemical methods.